In today’s rapidly evolving health, wellness, and industrial technology sectors, ultraviolet (ultravioleta) light devices have emerged as powerful tools with applications spanning medical therapy, water purification, air disinfection, surface sterilization, and advanced material curing. As global demand surges—accelerated by heightened awareness of hygiene and non-invasive therapeutic solutions—brands and businesses are increasingly turning to specialized UV light devices manufacturers and OEM suppliers to bring high-quality, compliant, and innovative products to market efficiently. This article explores why partnering with a professional OEM supplier is not just advantageous but essential for success in this dynamic industry.
Understanding UV Light Technology: Beyond the Basics
Ultraviolet light occupies the electromagnetic spectrum between visible light and X-rays, typically categorized into three primary bands:
- UVA (315–400 nm): Known for skin tanning and certain phototherapy applications; also used in curing adhesives and coatings.
- UVB (280–315 nm): Critical for medical phototherapy (p.ej., treating psoriasis, vitiligo, and eczema) and vitamin D synthesis.
- UVC (100–280 nm): Germicidal wavelength, highly effective at inactivating bacteria, viruses, and mold—making it indispensable for disinfection systems.
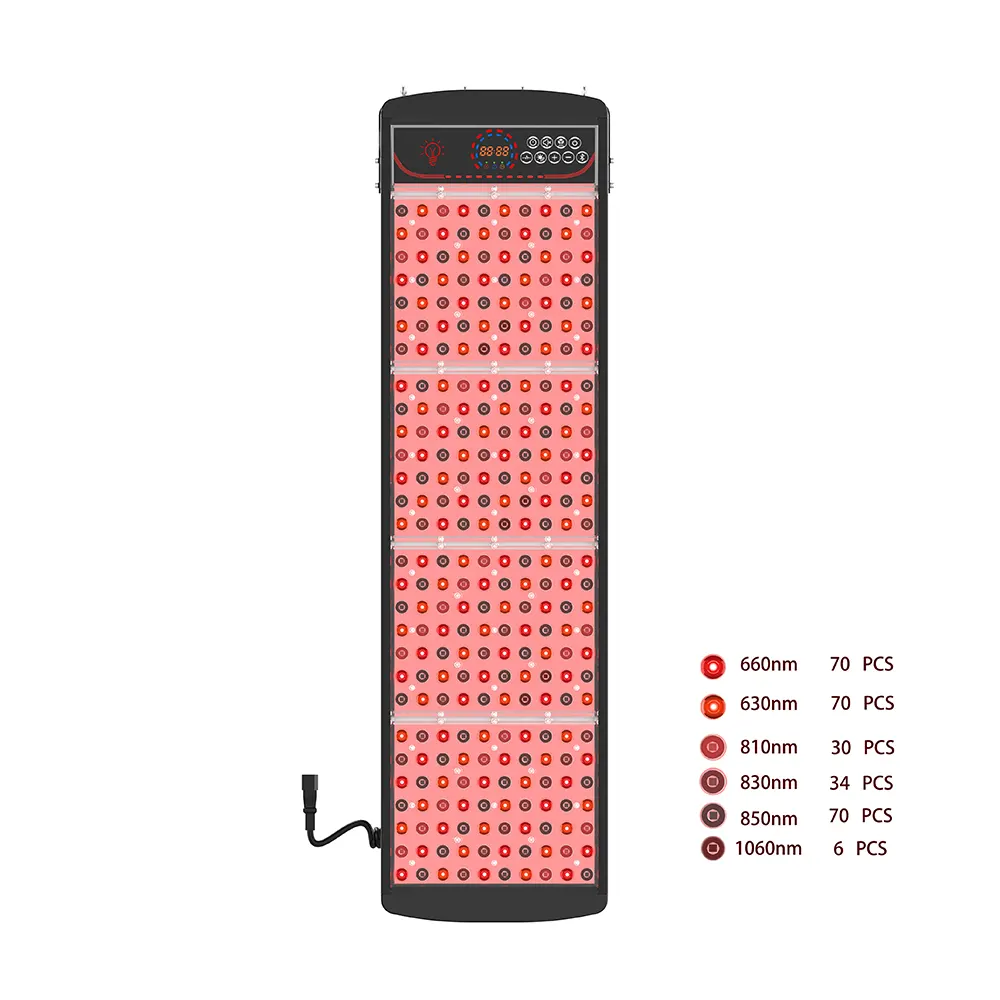
Advanced devices now integrate multi-wavelength arrays, combining UV with visible red light (630–660 nm) y infrarrojo cercano (NIR, 810–1060 nm) to unlock synergistic therapeutic benefits. Por ejemplo, a professional phototherapy panel might deploy nine distinct wavelengths to address skin conditions, accelerate wound healing, reducir la inflamación, and support cellular regeneration—all within a single treatment session.
Sin embargo, harnessing these wavelengths safely and effectively requires deep expertise in optical engineering, thermal management, electronic control systems, and biological interaction principles. This complexity underscores why brands benefit immensely from collaborating with an experienced OEM partner rather than attempting in-house development from scratch.
Why Choose an OEM Supplier for UV Devices?
Partnering with a dedicated UV light devices manufacturer offers strategic advantages that directly impact time-to-market, product quality, cumplimiento normativo, and scalability:
1. Accelerated R&D and Time-to-Market
A seasoned OEM supplier brings established R&D frameworks, pre-validated component libraries, and application-specific design templates. Instead of spending months (or years) prototyping and testing, brands can leverage the supplier’s existing knowledge base to fast-track development. Por ejemplo, a manufacturer with prior experience in medical-grade UVB panels can adapt proven circuit designs, LED binning strategies, and safety interlocks to a new product concept—reducing development cycles by 40–60%.
2. Cost Efficiency Through Scale and Expertise
OEM partners operate at scale, sourcing high-quality LEDs, conductores, optics, and housing materials at competitive volumes. They also optimize manufacturing workflows to minimize waste and rework. By outsourcing production, brands avoid capital expenditures on specialized equipment, cleanroom facilities, or hiring niche engineering talent—converting fixed costs into variable, predictable expenses.
3. Regulatory Navigation and Certification Support
UV devices—especially those intended for medical, therapeutic, or disinfection use—face stringent regulatory requirements globally (FDA, CE, RoHS, CEI 62471, MDSAP, etc.). A reputable OEM supplier maintains in-house compliance teams familiar with documentation, testing protocols, and submission processes. They can guide partners through certification pathways, conduct pre-compliance testing, and ensure designs meet electromagnetic compatibility (CEM), photobiological safety, and electrical safety standards from day one.
4. Quality Assurance and Reliability
Professional manufacturers implement rigorous quality management systems (often ISO 13485 for medical devices) covering incoming inspection, in-process controls, and final audit testing. Key parameters like irradiance uniformity, precisión de la longitud de onda, thermal stability, and operational lifespan are validated under real-world conditions. This diligence translates to fewer field failures, stronger brand reputation, and higher customer satisfaction.
Core Capabilities of a Professional UV Device Manufacturer
When evaluating potential OEM partners, look for these foundational capabilities:
🔹 Advanced Optical Design: Precision lensing, reflector engineering, and beam homogenization to ensure uniform irradiance across the treatment area—critical for both therapeutic efficacy and disinfection reliability.
🔹 Thermal Management Expertise: High-power UV LEDs generate significant heat. Effective heat sinking, active cooling solutions, and intelligent thermal throttling protect LED longevity and maintain consistent output.
🔹 Smart Control Systems: Integration of microcontrollers, touch interfaces, timer functions, dose calculators, and IoT connectivity enables user-friendly operation and data tracking—key differentiators in competitive markets.
🔹 Material Science & Ergonomía: Selection of UV-stable polymers, medical-grade aluminum, and ergonomic form factors ensures durability, seguridad, and user comfort across clinical, commercial, and home-use scenarios.
🔹 Scalable Production: Flexible manufacturing lines capable of handling low-volume pilot runs alongside high-volume mass production, with robust supply chain resilience to mitigate component shortages.
Quality Assurance & Global Certifications: Non-Negotiable Standards
Trust in UV devices hinges on verifiable safety and performance. Leading OEM suppliers invest heavily in certification portfolios that open global market access:
- CE & RoHS: Mandatory for European market entry, confirming electromagnetic compatibility and restriction of hazardous substances.
- Registro de la FDA & Listing: Essential for medical or therapeutic claims in the United States; includes facility registration and product listing.
- CEI 62471 Photobiological Safety: Evaluates risks from optical radiation (ultravioleta, visible, IR) to eyes and skin—critical for user safety documentation.
- MDSAP (Medical Device Single Audit Program): Streamlines regulatory audits across multiple jurisdictions (US, Canadá, Brazil, Japón, Australia).
- SAA/RCM: Required for electrical safety compliance in Australia and New Zealand.
A transparent supplier will provide test reports, certification copies, and factory audit summaries upon request—demonstrating commitment to accountability and continuous improvement.
Customization Services: Tailoring Solutions to Your Brand Vision
One of the greatest values an OEM partner delivers is end-to-end customization. Unlike off-the-shelf products, a collaborative OEM relationship enables:
✨ Configuración de longitud de onda: Select specific UV/visible/NIR wavelengths and power densities aligned with your target application—whether targeting bacterial inactivation (265 nm UVC), psoriasis treatment (311 nm UVB), or deep-tissue recovery (850 nm NIR).
✨ Factor de forma & Diseño Industrial: From handheld wands and wearable patches to large-area panels and ceiling-mounted systems, design flexibility ensures your product stands out aesthetically and functionally.
✨ Herrada & Embalaje: Custom logos, color schemes, user interfaces, manuales multilingües, and retail-ready packaging reinforce brand identity and enhance unboxing experience.
✨ Software & Conectividad: Develop companion apps for treatment tracking, remote firmware updates, usage analytics, or telehealth integration—adding significant value in digital health ecosystems.
✨ Etiqueta privada & White Label Options: Launch products under your brand name with minimal upfront investment, leveraging the supplier’s manufacturing excellence while focusing your resources on marketing and distribution.
Applications Across Industries: Where UV Innovation Thrives
The versatility of UV technology enables impactful applications across diverse sectors:
🏥 Médico & Dermatology: Targeted phototherapy for skin conditions, wound care acceleration, neonatal jaundice treatment, and surgical site disinfection.
🏠 Home Health & Bienestar: Consumer-friendly devices for acne management, anti-aging skincare, mood enhancement, and immune support—driving the fast-growing at-home therapy market.
🏢 Commercial Disinfection: HVAC-integrated UVC systems, upper-room germicidal fixtures, and mobile robots for hospitals, schools, offices, and hospitality venues.
💧 Water & Air Purification: Point-of-use water sterilizers, aquarium treatment units, and industrial air scrubbers leveraging UVC’s pathogen-inactivating properties.
🔬 Industrial & Scientific: UV curing for inks, adhesives, and 3D printing; fluorescence excitation in analytical instruments; and semiconductor processing.
A forward-thinking OEM supplier stays attuned to these evolving use cases, proactively developing platform technologies that can be adapted across verticals—maximizing your R&D ROI.
How to Select the Right OEM Partner: A Strategic Checklist
Choosing the ideal UV devices manufacturer requires due diligence. Prioritize partners who demonstrate:
✅ Proven Track Record: Request case studies, client references, and product portfolios relevant to your application domain.
✅ In-House Engineering: Avoid trading companies; seek suppliers with dedicated optical, electronic, mechanical, and software engineering teams under one roof.
✅ Transparent Communication: Clear project timelines, responsive technical support, and collaborative problem-solving are vital for smooth development cycles.
✅ IP Protection Policies: Formal NDAs, clear ownership agreements for custom designs, and secure data handling practices safeguard your innovations.
✅ Sustainability Commitment: Eco-conscious material selection, energy-efficient designs, and end-of-life recycling programs align with modern brand values and regulatory trends.
Conclusión: Building the Future of Light-Based Innovation Together
The UV light devices market represents a convergence of photonics, biology, and digital technology—a space ripe with opportunity for brands that act strategically. By partnering with a capable, certified, and customer-centric UV light devices manufacturer and OEM supplier, you gain more than a production vendor: you gain a co-innovator committed to transforming your vision into safe, eficaz, and market-leading products.
Whether you’re launching a breakthrough medical therapy device, scaling a disinfection solution for global distribution, or creating the next generation of wellness technology, the right OEM alliance provides the technical depth, regulatory agility, and manufacturing excellence to turn ambition into impact. In an industry where precision, seguridad, and performance are non-negotiable, collaboration isn’t just smart business—it’s the foundation of lasting success.