The hair restoration industry is experiencing unprecedented growth. As consumers increasingly seek non-invasive, drug-free solutions for hair loss, Terapia laser a basso livello (LLLT) devices have moved from clinical obscurity to mainstream wellness essentials. Per gli imprenditori, dermatologists, e marchi del benessere, this shift presents a lucrative opportunity. Tuttavia, entering this market requires more than just a logo; it demands a product that is safe, efficace, and legally compliant.
This is where the search for a “private label FDA registered hair growth laser cap manufacturer” becomes critical. Partnering with a manufacturer who understands the complexities of FDA regulations, laser safety, and medical device quality assurance is the foundation of a successful brand.
Presso Pureluxtech, we specialize in bridging the gap between advanced medical engineering and consumer-friendly design. As a leading OEM/ODM partner headquartered in Shenzhen, we empower brands to launch compliant, high-performance laser hair growth caps with confidence. This guide explores what it means to work with an FDA-registered manufacturer, the benefits of private labeling, and why Pureluxtech is the strategic choice for your next product launch.

Understanding FDA Compliance: Registration vs. Liquidazione
1. FDA Facility Registration
- Cosa significa: The factory is known to the FDA and subject to inspection.
- Pureluxtech Status: Our Shenzhen facility is fully registered and operates under an Sistema di gestione della qualità certificato ISO 13485, ensuring international medical device standards are met.
2. FDA 510(k) Liquidazione
- Cosa significa: The specific laser cap model is cleared for safety and intended use.
- Pureluxtech Support: We offer FDA 510(k)-cleared platforms that private label partners can leverage. Forniamo i file tecnici necessari, rapporti di prova, and documentation to support your brand’s listing and regulatory compliance.
3. Why This Distinction Matters for Your Brand
The Strategic Advantages of Private Labeling
Choosing a private label model over building a factory from scratch offers significant competitive advantages, especially in the highly technical LLLT sector.
1. Speed to Market
Developing a laser device from scratch involves years of R&D, clinical testing, and regulatory submission. Private labeling allows you to leverage existing, proven platforms.
- Vantaggio Pureluxtech: You can launch in 3-6 months instead of 2-3 years by utilizing our pre-engineered, compliance-ready designs.
2. Efficienza dei costi
Building an SMT line, optical lab, and clean room requires millions in capital expenditure. Private labeling converts fixed costs into variable costs.
- Vantaggio Pureluxtech: No upfront tooling costs for standard models. You invest in inventory and branding, not infrastructure.
3. Regulatory Risk Mitigation
Navigating FDA, CE, and IEC laser safety standards is complex. Mistakes can be costly.
- Vantaggio Pureluxtech: We handle the heavy lifting of compliance documentation. Our regulatory team ensures every unit shipped meets the standards required for your target market (US, Unione Europea, Regno Unito, Australia).
4. Brand Focus
Outsourcing manufacturing allows you to focus on what you do best: marketing, customer service, and distribution.
- Vantaggio Pureluxtech: We act as your silent partner, handling production, controllo di qualità, and logistics while you build brand equity.
Pureluxtech: Your Trusted Private Label Partner
Our Core Capabilities for Private Label Brands
|
Caratteristica
|
Standard Pureluxtech
|
|---|---|
|
Conformità normativa
|
FDA Facility Registered & ISO 13485 Certificato. 510(k)-cleared platforms available for private label.
|
|
Laser Technology
|
Genuine 650nm Medical Lasers. Combined with 630/850nm LEDs for hybrid efficacy.
|
|
Personalizzazione
|
Full Private Label. Logo, confezione, manual, app interface, and even hardware tweaks.
|
|
Controllo di qualità
|
6-Step QC Process. 100% laser power testing, 48-hour aging, drop testing, e controlli di sicurezza.
|
|
Capacità produttiva
|
Scalable Lines. From pilot batches (low MOQ) to mass production (10k+ unità/mese).
|
|
Catena di fornitura
|
Integrazione verticale. Direct sourcing of laser diodes, batteries, and medical-grade plastics.
|
|
Supporto
|
7Servizio globale ×24. Dedicated account managers and technical support teams.
|
|
Logistica
|
Global Shipping. DDP options, 3PL partnerships, and hazardous material (batteria) conformità.
|
|
R&D
|
Continuous Innovation. New models, funzionalità intelligenti, and ergonomic improvements released annually.
|
|
Traccia di registrazione
|
10,000+ Clienti. Proven success with startups, cliniche, and enterprise brands.
|
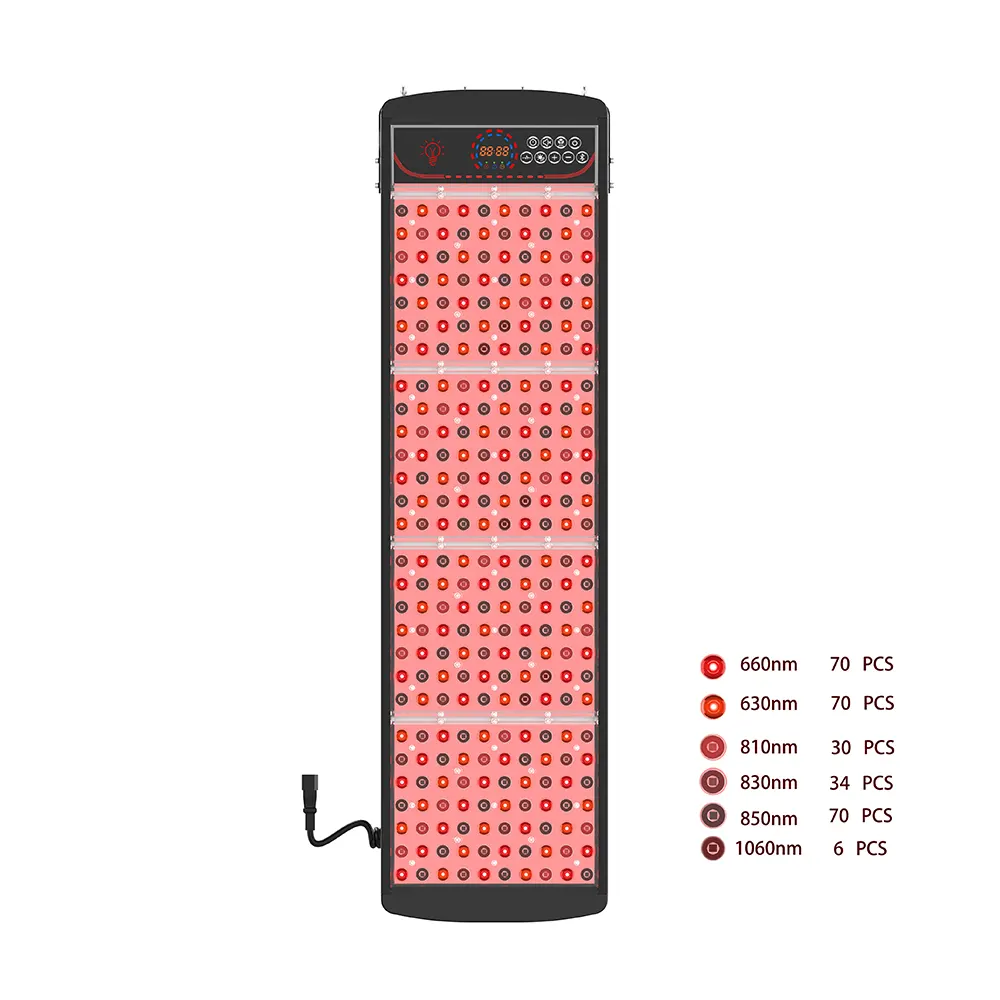
Our Private Label Laser Cap Series
- Entry-Level (80-110 Lasers): Affordable, portatile, ideal for preventative care and mass market retail.
- Professionale (150-200 Lasers): Balanced power and coverage, perfect for DTC brands and clinics.
- Premio (272+ Lasers): Maximum density for severe hair loss, positioned as a luxury medical device.
- Smart Connected: Bluetooth app integration for treatment tracking and customer retention.
The Private Label Process: From Concept to Launch
Fare un passo 1: Consultazione & Strategia
Fare un passo 2: Product Selection & Personalizzazione
- Marchio: We apply your logo via laser etching, printing, or molding.
- Confezione: Custom box design, inserti, and user manuals in your brand voice.
- Software: Custom app skins and welcome screens for smart devices.
Fare un passo 3: Conformità & Documentazione
- FDA Facility Registration numbers.
- 510(k) reference letters (where applicable).
- CEI 60825-1 Laser Safety reports.
- UN38.3 Battery Safety certificates.
- CE/UKCA/RoHS documentation for international markets.
Fare un passo 4: Campionamento & Validazione
Fare un passo 5: Produzione & Consegna
Technical Excellence: What Makes Our Laser Caps Different
1. Genuine 650nm Laser Diodes
2. Hybrid Light Technology
3. Sicurezza & Gestione termica
4. Design ergonomico
Risks of Sourcing from Non-Compliant Manufacturers
The allure of low costs can be dangerous. Sourcing from a manufacturer who is not truly FDA registered or lacks laser expertise carries significant risks:
- Customs Seizure: Devices lacking proper FDA/CE documentation can be confiscated at the border, resulting in total loss of inventory.
- Legal Liability: Selling non-compliant laser devices exposes your brand to lawsuits if users experience eye damage or skin burns.
- Ineffective Products: Cheap diodes often emit incorrect wavelengths (per esempio., 670nm instead of 650nm), rendering the device useless for hair growth. This leads to high return rates and brand damage.
- Battery Hazards: Uncertified lithium batteries pose fire risks during shipping and use, violating safety laws.
Pureluxtech eliminates these risks through transparency, certificazioni verificate, and a commitment to long-term partnership. We don’t just sell boxes; we sell peace of mind.
Launch Your Hair Restoration Brand with Pureluxtech
The demand for effective, non-invasive hair loss solutions is at an all-time high. Consumers are looking for trusted brands that offer safe, clinically-backed technology. By partnering with a private label FDA registered hair growth laser cap manufacturer like Pureluxtech, you position your brand for success from day one.
We combine medical-grade engineering with consumer-centric design to create products that deliver real results. Whether you are a startup looking for a low-MOQ entry point or an established brand seeking to expand your portfolio, we have the expertise to scale with you.
Domande frequenti (Domande frequenti)
UN: SÌ. We use medical-grade laser diodes with a tolerance of ±5nm. We provide spectrometer test reports for every production batch to verify wavelength accuracy.
UN: Our laser caps use genuine 650nm laser diodes which penetrate deeper into the follicle than LEDs. We often combine lasers with LEDs in a hybrid design to maximize both depth and coverage for superior results.
UN: SÌ. We offer platforms that are FDA 510(k) cleared or ready for submission. We provide all necessary technical files, safety tests (CEI 60825-1), and documentation to support your registration.
UN: We use high-quality lithium polymer batteries with UN38.3 certification. Our devices include intelligent BMS (Battery Management Systems) to prevent overcharging, overheating, and short circuits.
UN: We offer flexible MOQs to support startups and established brands alike. Contact us for a quote based on your specific customization requirements (conteggio dei diodi, progetto, caratteristiche).