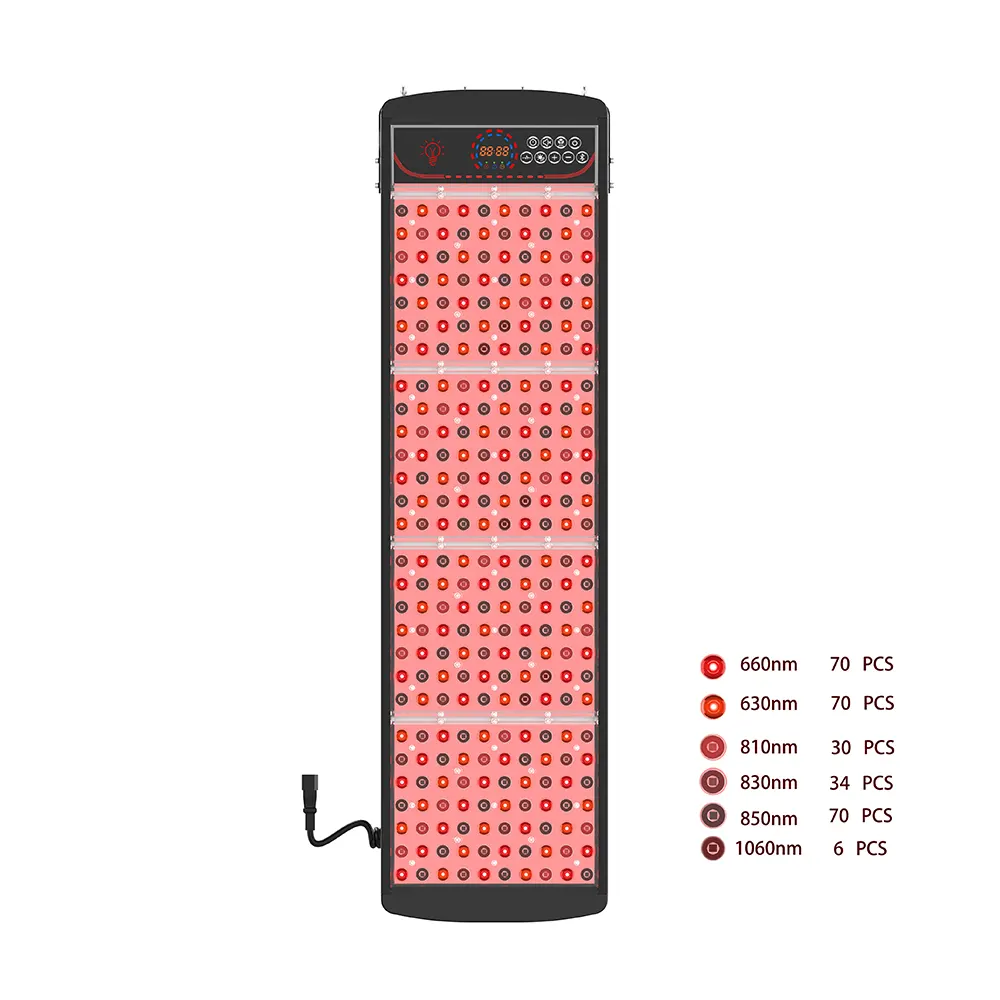
Entering the USA and EU beauty tech markets offers immense opportunity—but also significant regulatory complexity. For brands targeting health-conscious consumers in these regions, compliance isn’t optional; it’s the foundation of trust, liability protection, 장기적인 확장성. That’s why sourcing from an OEM FDA/CE certified red light eye mask manufacturer for USA/EU market isn’t just a procurement decision—it’s a strategic safeguard that accelerates market entry, minimizes legal risk, and builds consumer confidence from day one.
This guide breaks down what true certification means, how regulatory pathways differ between the USA and EU, and why partnering with a compliance-first OEM manufacturer like Pureluxtech transforms regulatory complexity into competitive advantage.
인증이 중요한 이유: Beyond the Logo
In the wellness and beauty device space, “FDA 등록” 또는 “CE marked” claims are frequently misused. Understanding the distinction is critical:
🔹 FDA Registration (미국): For non-invasive red light therapy devices marketed for general wellness (예를 들어, “promotes relaxation,” “supports skin appearance”), manufacturers typically register their facility with the FDA under 21 CFR Part 807 and list devices via the FURLS system. This is not the same as FDA “approval,” which applies to medical devices requiring 510(케이) 정리. 하지만, proper registration demonstrates transparency, manufacturing accountability, and adherence to Quality System Regulation (QSR) guidelines.
- Low Voltage Directive (LVD) 2014/35/EU – Electrical safety
- 전자기 호환성 (EMC) Directive 2014/30/EU – Interference control
- RoHS Directive 2011/65/EU – Restriction of hazardous substances
- REACH Regulation – Chemical safety in materials
- General Product Safety Regulation (GPSR) – Consumer protection

USA vs. EU: Key Regulatory Differences for Beauty Tech
|
Requirement
|
USA Market
|
EU Market
|
|---|---|---|
|
Primary Framework
|
FDA Wellness Device Registration
|
CE Marking under Multiple Directives
|
|
라벨링
|
English-only; FDA facility registration number optional but recommended
|
Multilingual (per destination country); CE logo + Notified Body ID if applicable
|
|
기술 문서
|
Device listing, manufacturing QSR, basic safety testing
|
Full Technical File, EU Declaration of Conformity, risk management report
|
|
Post-Market Obligations
|
Adverse event reporting (if medical claims made)
|
Vigilance reporting, UDI registration (EUDAMED), authorized EU representative
|
|
Testing Standards
|
UL/ETL for electrical safety; FCC for wireless
|
EN 62471 (photobiological safety), EN 60601-1 (if medical intent), EMC testing
|
퓨어럭스텍: Compliance Engineered Into Every Unit
As a dedicated OEM FDA/CE certified red light eye mask manufacturer for USA/EU market, Pureluxtech builds regulatory readiness into our development process—not as an afterthought, but as a core design principle.
In-House Regulatory & Testing Infrastructure
- Dedicated Compliance Team: Engineers and documentation specialists trained in FDA QSR, EU MDR/IVDR frameworks, and international electrical safety standards.
- Accredited Testing Partnerships: We collaborate with ISO 17025-accredited labs for photobiological safety (IEC 62471), EMC, 열의, and battery safety testing—providing full test reports for your technical files.
- 파장 & Irradiance Validation: Every batch undergoes spectral calibration to ensure output remains within therapeutic and safe limits, documented for regulatory submissions.
End-to-End Documentation Support
✅ EU Technical File templates with risk assessments (ISO 14971)
✅ Declarations of Conformity (DoC) signed and dated
✅ Material Safety Data Sheets (MSDS) and biocompatibility reports (ISO 10993)
✅ Multilingual labeling files compliant with FDA 21 CFR 801 and EU GPSR
✅ UDI assignment guidance for EU EUDAMED registration
Market-Specific Customization
- USA Focus: English-only packaging, UL-certified USB-C chargers, FCC ID for Bluetooth models, wellness-focused marketing language aligned with FDA guidance.
- EU Focus: Multilingual manuals (EN, DE, FR, ES, IT), CE logo placement per directive requirements, EU Authorized Representative designation support, eco-compliant packaging per EPR regulations.
Risk Mitigation: Protecting Your Brand from Regulatory Pitfalls
⚠️ Marketplace Suspension: 아마존, 쇼피파이, and TikTok Shop increasingly require proof of compliance. Missing documentation can trigger listing removal or account penalties.
⚠️ Liability Exposure: If a non-compliant device causes injury or fails safety tests, brands—not manufacturers—often bear legal and reputational consequences.
⚠️ Rebranding Costs: Retrofitting packaging, labels, or firmware post-production is far more expensive than building compliance in from the start.
The OEM Partnership Process: Compliance-First, Speed-Optimized
- Regulatory Consultation: Share your target countries, marketing claims, and sales channels. We map requirements to device specs, 라벨링, and documentation needs.
- Pre-Compliance Prototyping: Receive functional samples with preliminary test reports (optical, electrical, 열의). Validate fit, 성능, and user experience before finalizing compliance documentation.
- Documentation Finalization: Approve technical files, Declarations of Conformity, labeling artwork, and multilingual manuals. We coordinate third-party testing if required for your specific claims or market.
- Pre-Production Validation: A pilot batch undergoes full compliance verification: EMC scan, photobiological safety check, drop/aging tests. Reports are shared for your records or marketplace submission.
- 양산 & Audit-Ready QC: Automated assembly with batch traceability. Every unit is functionally tested; random samples undergo full compliance re-verification.
- 출시 후 지원: Ongoing assistance with regulatory updates, marketplace compliance inquiries, firmware revisions, and expansion to new regions (UKCA, 호주, 캐나다).
Who Needs a Certified OEM Partner?
🇪🇺 EU Market Entrants: Brands expanding into Germany, France, 이탈리아, or pan-European marketplaces requiring CE documentation and multilingual compliance.
🌐 Global Scalability Seekers: Companies planning to launch in both regions simultaneously, benefiting from unified engineering and parallel documentation workflows.
🏥 Professional Channel Suppliers: Distributors selling to clinics, 스파, or dermatology practices where liability protection and clinical-grade documentation are non-negotiable.