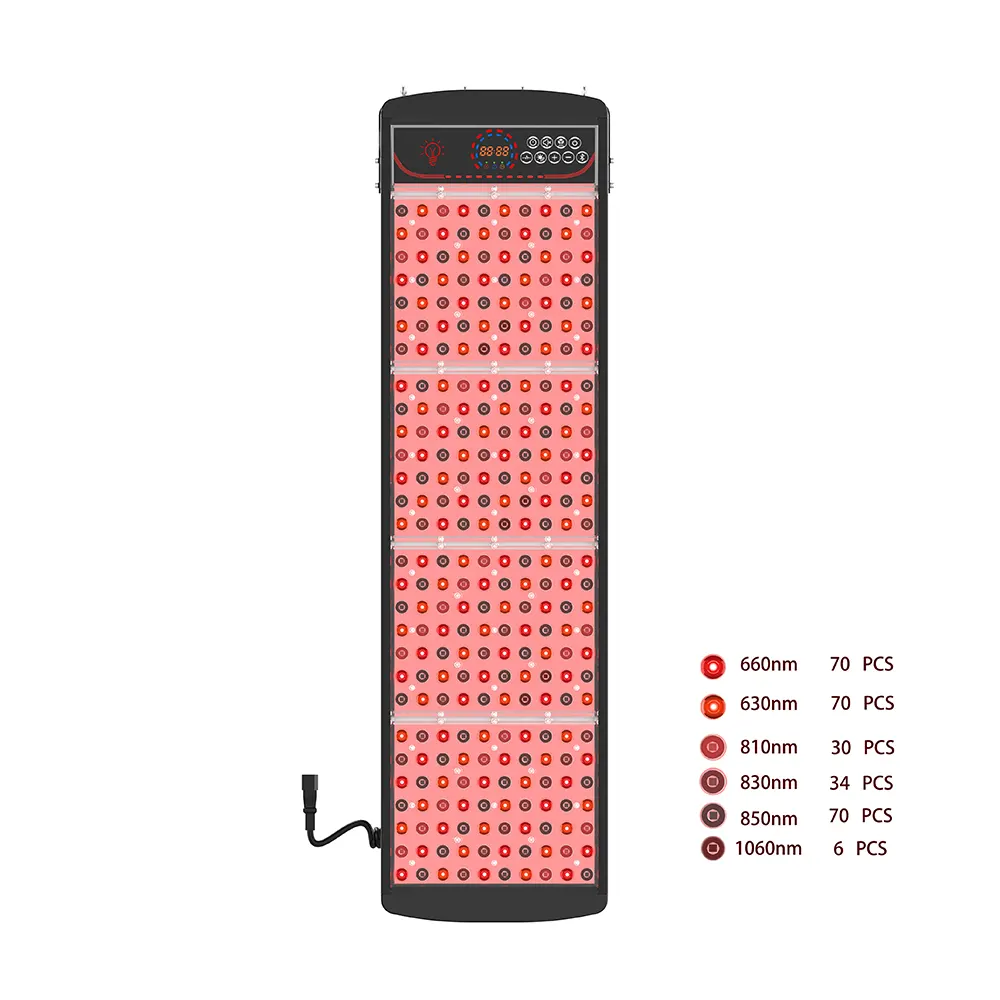
The global UV light therapy market is expanding at an unprecedented pace, driven by rising dermatological conditions, growing consumer adoption of at-home wellness devices, and increasing clinical validation of phototherapy protocols. ສໍາລັບຍີ່ຫໍ້, ຕົວແທນຈໍາຫນ່າຍ, and healthcare innovators, success in this dynamic sector depends less on having a great idea and more on executing that idea with precision-engineered, ສອດຄ່ອງ, and commercially viable hardware. This is where a strategic OEM partnership becomes transformative. Pureluxtech, a vertically integrated manufacturer specializing in UV and phototherapy light devices, offers more than component supply—it delivers end-to-end engineering excellence, ຄວາມຊໍານານລະບຽບການ, and scalable production that empowers partners to lead, not just compete.
Why Pureluxtech Stands Out in the UV OEM Landscape
Not all manufacturers are created equal. Pureluxtech distinguishes itself through a combination of technical depth, operational scale, and customer-centric collaboration:
🔹 Vertical Integration & In-House Expertise: With a 13,000m² R&D and manufacturing facility, 60+ dedicated phototherapy engineers, ແລະ 300+ patents in optical design, thermal management, and smart control systems, Pureluxtech controls the entire value chain—from LED binning and optical simulation to firmware development and final calibration. This eliminates dependency on third-party assemblers and ensures consistent quality, faster iteration, and protected IP.
🔹 Dual-Market Certification Mastery: Whether targeting FDA-regulated medical devices in the U.S. or CE-marked products under EU MDR, Pureluxtech embeds compliance into the design phase. Their in-house regulatory team supports technical file preparation, pre-compliance testing (IEC 62471, IEC 60601-1, EMC), and liaison with notified bodies—accelerating time-to-market while de-risking submissions.
🔹 Platform-Based Engineering for Speed & Flexibility: Rather than building every product from scratch, Pureluxtech leverages modular optical engines, standardized control architectures, and configurable firmware frameworks. ນີ້ “platform approach” allows partners to customize wavelength profiles, irradiance levels, form factors, and connectivity features without sacrificing reliability or extending development timelines by 12+ ເດືອນ.
🔹 Global Support Infrastructure: With headquarters in China and a U.S. commercial office in Irvine, California, Pureluxtech provides localized engineering support, responsive communication across time zones, and logistics coordination for North American, European, and emerging market deployments.
End-to-End Partnership Model: From Concept to Commercial Scale
Pureluxtech structures engagements as collaborative innovation journeys, not transactional supply agreements. The workflow is transparent, milestone-driven, and designed for scalability:
ໄລຍະ 1: ການຄົ້ນພົບ & ຂໍ້ມູນຈໍາເພາະ
Joint workshops define clinical targets, user personas, regulatory pathways, irradiance requirements, and commercial constraints. Pureluxtech’s application engineers help translate therapeutic goals into technical specifications.
ໄລຍະ 2: Optical & Mechanical Prototyping
Using ray-tracing software, thermal simulation tools, and rapid prototyping facilities, Pureluxtech delivers functional alpha units for hands-on validation. Iterative testing ensures spectral accuracy, dose uniformity, and ergonomic usability.
ໄລຍະ 3: Firmware & Smart Integration
Custom control logic, dose algorithms, Bluetooth/Wi-Fi connectivity, and API endpoints are developed to align with your brand’s digital ecosystem—whether a consumer mobile app, telehealth platform, or clinic management software.
ໄລຍະ 4: Pre-Compliance Validation
In-house photobiological, electrical safety, and EMC testing de-risk formal certification submissions. Pureluxtech provides complete test reports, risk management files (ISO 14971), and technical documentation ready for audit.
ໄລຍະ 5: Pilot Production & Field Testing
Small-batch manufacturing enables real-world validation with clinicians, beta users, or retail partners. Feedback informs final design refinements before mass production.
ໄລຍະ 6: ການຜະລິດມະຫາຊົນ & Global Logistics
Scalable output with serialized tracking, 100% irradiance calibration, and quality audits ensures consistency. Pureluxtech coordinates freight, customs documentation, and regional warehousing support.
ໄລຍະ 7: Post-Launch Partnership
Firmware updates, spare parts logistics, warranty fulfillment, and continuous improvement cycles based on field data ensure long-term product success and brand reputation protection.
ຄວາມສາມາດໃນການປັບແຕ່ງ: Tailoring Technology to Your Brand Vision
- Spectral Precision: Configure narrowband UVB (311 ນມ) for psoriasis/vitiligo, UVA1 (340–400 nm) for anti-inflammatory therapy, or hybrid UV + red/NIR arrays for synergistic skin renewal.
- Form Factor Innovation: Design handheld wands, wearable patches, facial panels, full-body stands, or modular add-ons that align with your product strategy and user experience goals.
- ການເຊື່ອມຕໍ່ອັດສະລິຍະ: Embed treatment logging, dose tracking, telehealth integration, OTA updates, and AI-assisted protocol recommendations to differentiate in the connected health market.
- ການສ້າງຍີ່ຫໍ້ & Industrial Design: Custom housing finishes, UI language, logo placement, retail-ready packaging, and multilingual documentation reinforce brand identity and premium positioning.
- Private-Label Enablement: From hardware to software to marketing collateral, Pureluxtech supports fully white-labeled solutions that let you focus on brand building while they handle technical execution.
Commercial Impact: Measurable Advantages for Partners
Brands that partner with Pureluxtech realize tangible business outcomes:
📈 Accelerated Time-to-Market: Platform-based engineering and pre-validated compliance pathways reduce development cycles by 40–60%, enabling first-mover advantage in fast-moving categories.
💰 Optimized Unit Economics: Vertical integration, bulk LED sourcing, and efficient production workflows deliver competitive pricing without sacrificing quality—improving margins or enabling aggressive retail positioning.
🛡️ Reduced Regulatory Risk: Proactive compliance engineering minimizes costly redesigns, certification delays, or post-market enforcement actions.
🔄 Scalable Growth Support: MOQ ທີ່ມີຄວາມຍືດຫຍຸ່ນ, phased production scaling, and global logistics coordination allow partners to validate markets before committing to large inventory investments.
🌍 Global Market Readiness: Multi-region certification expertise (FDA, CE, TGA, PMDA) and localization support simplify expansion into high-value international markets.
Future-Ready Innovation: Anticipating Market Evolution
Pureluxtech invests in R&D to stay ahead of emerging trends:
- Hybrid Light Architectures: Combining UV with visible red, NIR, and blue wavelengths for multi-modal skin health platforms.
- AI-Driven Personalization: Machine learning algorithms that adapt dose protocols based on user feedback, skin response, and treatment history.
- Sustainable Design: Mercury-free components, energy-efficient drivers, and recyclable materials aligned with global ESG mandates.
- Telehealth Integration: Cloud-connected devices enabling remote clinician oversight, electronic health records syncing, and subscription-based care models.
By partnering with an innovation-focused manufacturer, brands future-proof their portfolios against technological disruption.
ສະຫຼຸບ: Your Strategic Advantage in Phototherapy Innovation
In the competitive UV light therapy market, execution determines success. A strategic OEM partnership with Pureluxtech provides the technical mastery, regulatory agility, production scalability, and collaborative mindset required to transform visionary concepts into market-leading products. Whether launching a breakthrough medical device, scaling a DTC wellness brand, or equipping a global clinic network, Pureluxtech empowers partners to deliver safe, ມີປະສິດທິພາບ, and commercially compelling solutions with confidence.
In an industry where precision, compliance, and user trust are non-negotiable, choosing the right manufacturing ally isn’t just a procurement decision—it’s the foundation of sustainable growth and category leadership.