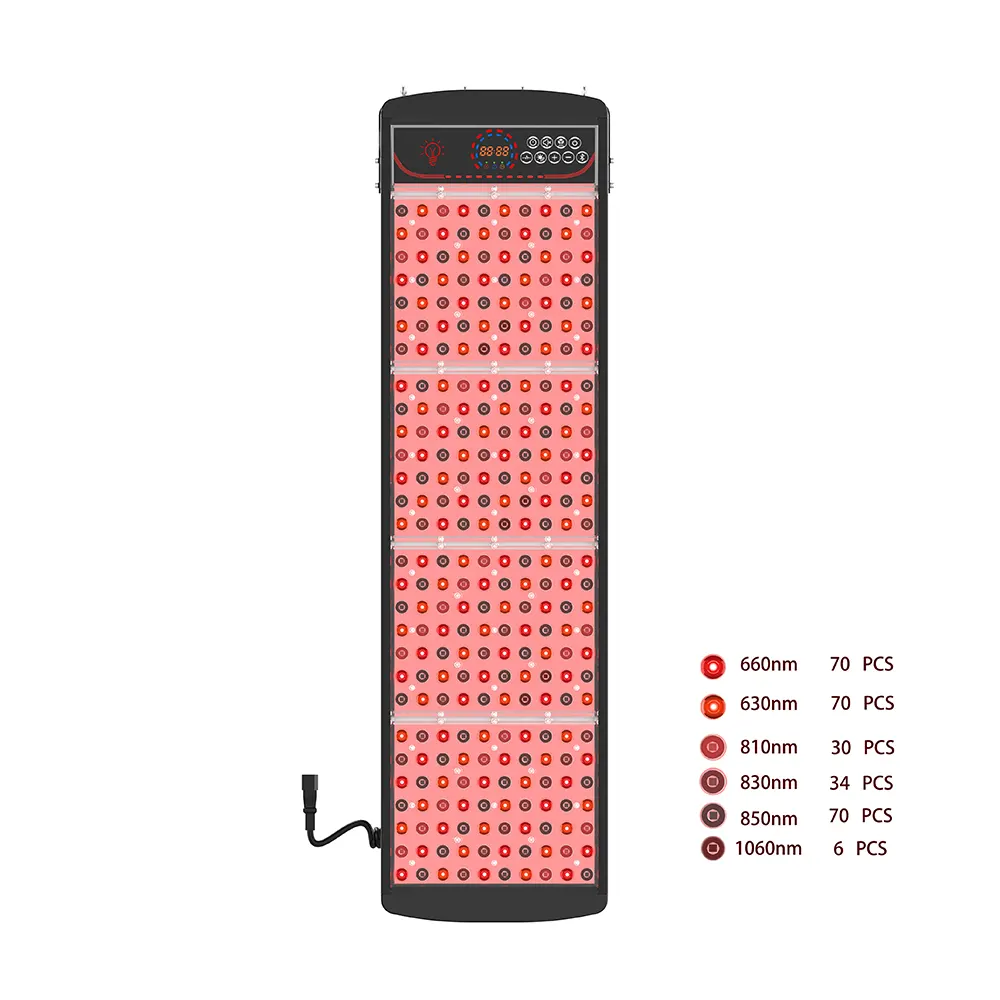
The therapeutic potential of photobiomodulation (PBM) hinges on precise wavelength delivery. Among the spectrum of available options, **660nm **(visible red) and **850nm **(nabij-infrarood) have emerged as the gold-standard combination for comprehensive wellness applications. For businesses seeking to source, distribute, or private-label these devices, selecting the right 660nm 850nm red light therapy panel manufacturer is the most critical decision you will make.
This guide explores what defines a qualified manufacturer for dual-wavelength panels, outlines essential technical and operational criteria, and explains why Pureluxtech has become a trusted partner for brands and distributors worldwide in 2026.
Waarom 660 nm + 850nm? The Science Behind the Standard Combination
Before evaluating manufacturers, it is essential to understand why this specific wavelength pairing dominates the market:
|
Golflengte
|
Primary Penetration
|
Key Therapeutic Benefits
|
|---|---|---|
|
**660nm **(Zichtbaar rood)
|
1–3 mm (epidermis/dermis)
|
Collageen stimulatie, huidverjonging, acne-beheer, surface wound healing
|
|
**850nm **(Nabij-infrarood)
|
20–40mm (spier, gewricht, nerve)
|
Deep-tissue repair, pijnverlichting, herstel van de spieren, anti-inflammatory effects
|

Core Capabilities of a Qualified 660nm/850nm Manufacturer
When vetting potential partners, prioritize these foundational capabilities:
1. Wavelength Precision and Verification
- Behouden ±5 nm tolerantie for peak 660nm and 850nm outputs
- Provide batch-specific spectrometer reports verifying wavelength accuracy
- Use premium LED chips (bijv., Epister, San’an) with tight binning tolerances
✅ Pureluxtech-standaard: All E Series and Esplus Series panels undergo spectrometer verification at the batch level, ensuring 660nm and 850nm outputs remain within clinically relevant tolerances.
2. Verified Irradiance at Treatment Distance
- Documentation of irradiance measured at **6 inch **(15 cm)—the standard treatment distance
- Target ≥100 mW/cm² for home-use panels; ≥150 mW/cm² for clinical-grade units
- Irradiance mapping showing uniform output across the panel surface
|
Pureluxtech Model
|
Irradiance at 6″
|
Best Application
|
|---|---|---|
|
≥230 mW/cm²
|
Localized facial or joint treatment
|
|
|
≥240 mW/cm²
|
Full-torso or limb recovery
|
|
|
≥200 mW/cm²
|
Large-area clinical or commercial use
|
3. Advanced LED Architecture
- Standard Single-Wavelength LEDs: Reliable and cost-effective for basic applications
- Dual-Core Dual-Wavelength Chips: As featured in Pureluxtech’s Esplus Series, these emit both 660nm and 850nm light simultaneously from a single housing, increasing energy density without proportional heat increase
- Optical Lens Design: 30° lenses for focused deep penetration; 60° lenses for wider surface coverage
4. Thermal Management and Build Quality
- Aircraft-grade aluminum housing with optimized fin structures for passive cooling
- Thermal interface materials that efficiently transfer heat from LEDs to heatsink
- Surface temperature control maintaining safe touch levels (<50°C) even after extended operation
💡 Pureluxtech Advantage: E Series and Esplus Series panels feature precision-machined aluminum chassis with passive thermal management, inschakelen 50,000+ hours of stable operation without noisy cooling fans.
5. Driver Quality and Control Flexibility
- Constant current regulation maintaining brightness despite voltage fluctuations
- **High efficiency **(>90%) reducing wasted energy as heat
- Pulse mode support enabling therapeutic frequencies (10Hz, 40Hz) without waveform distortion
- Independent channel control allowing users to toggle 660nm-only, 850nm-only, or combo modes
Manufacturing Processes That Ensure Consistency
🔹 Quality Control Protocols
|
QC Step
|
Purpose
|
Frequentie
|
|---|---|---|
|
Spectrometer verification
|
Confirm wavelength accuracy
|
Per batch
|
|
Irradiance mapping
|
Validate power distribution
|
Per unit or random sampling
|
|
Aging test (24–48 hour burn-in)
|
Identify early failures
|
100% of units
|
|
Thermal imaging
|
Detect hotspots or cooling issues
|
Random sampling
|
|
Drop/vibration test
|
Ensure shipping durability
|
Per production run
|
🔹 Certification and Compliance Support
- CE, RoHS, FCC: Basic requirements for EU and North American distribution
- FDA-registratie: Facility registration and device listing for U.S. geloofwaardigheid van de markt
- Technical Files: Documentation supporting safety and performance claims for clinical or insurance purposes
✅ Pureluxtech Commitment: Provides CE, RoHS, FCC documentation and FDA registration guidance for commercial partners, with technical files available to support accreditation discussions.
OEM/ODM Capabilities for Brand Differentiation
🔹 Customization Depth
- Golflengteverhoudingen: Adjust red-to-NIR ratios for skin-focused vs. pain-relief positioning
- Power Scaling: Offer models from 300W (home use) to 4500W+ (clinical arrays)
- Besturingssystemen: Custom timers, pulsmodi, dimmen, or app connectivity
- Branding: Lasergraveren, custom colors, verpakking ontwerp, meertalige handleidingen
- Mounting Solutions: Stellage, muurbevestiging, or ceiling-mount options tailored to use case
🔹 Scalable Engagement Models
- Low-MOQ Programs: Start with 50–100 units to validate demand before scaling
- Phased Customization: Begin with branding-only options, then advance to technical customization
- Dedicated Support: Bilingual account managers and engineering contacts for smooth communication
💡 Pureluxtech OEM Support: Offers flexible customization across all product lines, with low MOQs for startups and scalable solutions for enterprise clients.
Why Pureluxtech Stands Out as a 660nm/850nm Specialist
Advanced Product Platforms
- **E-serie **(ES300 to ES4500): Scalable power options with precise 660nm/850nm combinations, hoge bestraling (≥230–300 mW/cm² at 6″), and robust aluminum housing. Ideal for brands targeting home wellness or clinical markets.
- Esplus Series: Features dual-core dual-wavelength technology and extended NIR options (tot 1060nm), enabling advanced therapeutic differentiation for premium or medical-adjacent brands.
Verified Performance Documentation
Long-Term Reliability Backed by Warranty
Partnership-Focused Support
Practical Checklist: Evaluating a 660nm/850nm Manufacturer
Use this quick reference during your vendor assessment process:
- Request spectrometer reports confirming 660nm/850nm peaks within ±5nm tolerance
- Verify irradiance data measured at 6 inch, not just panel surface
- Review build quality: aluminum housing, passive cooling, industrial connectors
- Validate certifications: CE, RoHS, FCC, FDA registration documentation
- Assess warranty terms: length, dekkingsbereik, and RMA process clarity
- Evaluate OEM flexibility: wavelength tuning, power scaling, branding options
- Test communication responsiveness and technical knowledge during sales process
- Request references from existing clients in your target region
- Review total cost of ownership, not just unit price
- Confirm sample availability for pre-production testing
Final Takeaway: Precision Engineering Drives Therapeutic Results
Selecting a 660nm 850nm red light therapy panel manufacturer is fundamentally about choosing a partner who can deliver verified performance, naleving van de regelgeving, and scalable support. The right manufacturer ensures that your products provide real therapeutic value—building customer trust and protecting your brand reputation.
Pureluxtech embodies this standard. With advanced dual-wavelength engineering, batch-level quality verification, flexible OEM programs, and a 5-year warranty backed by responsive support, they provide the foundation businesses need to succeed in photobiomodulation.
Whether you are launching a new private-label brand, expanding a clinical product line, or entering the PBM market for the first time, partnering with a manufacturer that prioritizes both technical excellence and commercial collaboration turns your vision into a high-performance reality.