The skincare and dermatological wellness markets are undergoing a paradigm shift. Consumers and clinicians alike are moving beyond topical treatments and oral medications toward targeted, non-invasive light-based therapies. Ultraviolet (UV) skin therapy has emerged as a clinically validated modality for managing psoriasis, vitiligo, eczeem, cutaneous T-cell lymphoma, and certain inflammatory dermatoses.
As demand accelerates, brands are increasingly turning to custom UV skin therapy device OEM private label manufacturers to launch differentiated, meewerkend, and market-ready product lines without the capital risk of building internal R&D and production infrastructure. This article outlines how strategic OEM partnerships enable brands to harness phototherapy innovation while maintaining full control over identity, pricing, and market positioning.
Why Customization Is the New Competitive Edge in UV Skin Therapy
UV phototherapy is inherently condition-specific. Broad-spectrum approaches are rapidly being replaced by precision-engineered solutions that match wavelength, bestraling, behandelgebied, and safety protocols to distinct clinical or consumer needs. Customization allows brands to:
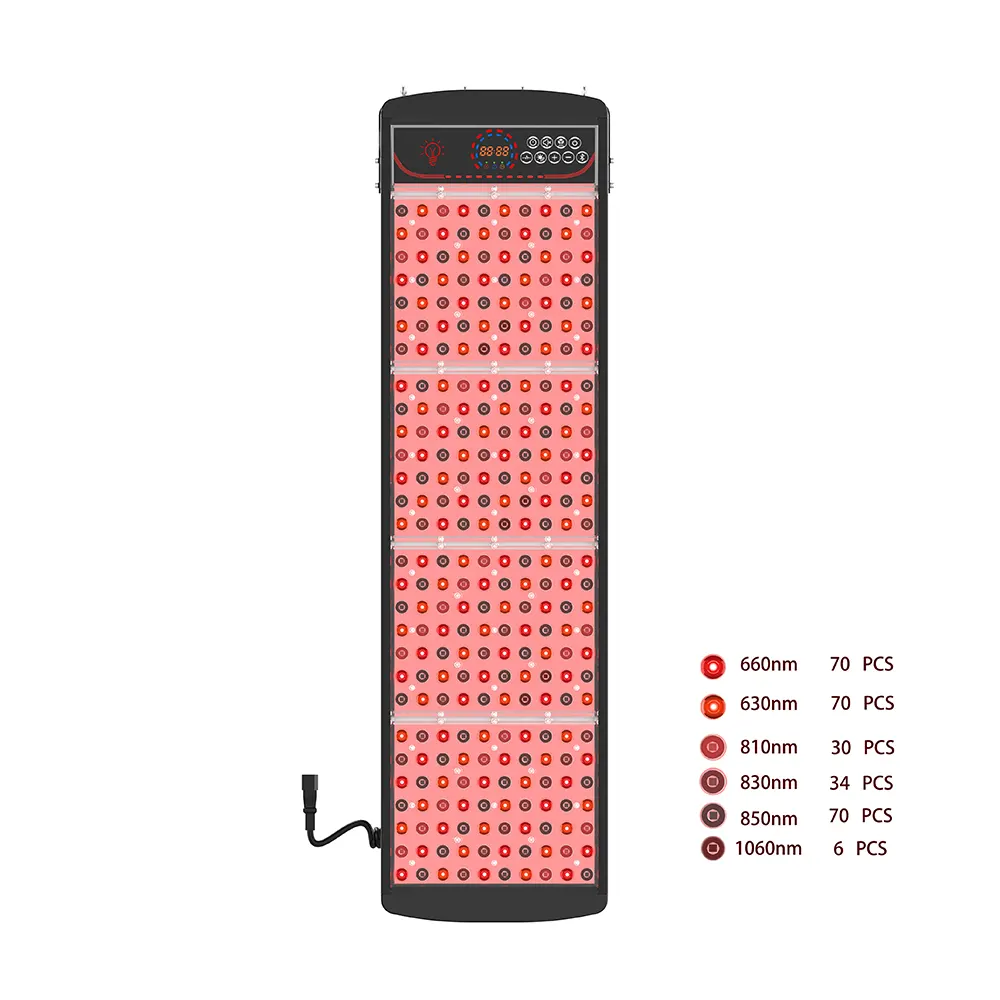
🔹 Target Specific Indications: Narrowband UVB (311–313 nm) for psoriasis and vitiligo, UVA1 (340–400 nm) for morphea and atopic dermatitis, or hybrid arrays combining UV with red/NIR wavelengths for synergistic anti-inflammatory and barrier-repair effects.
🔹 Optimize Treatment Geometry: Handheld wands for localized lesions, modular panels for joint areas, full-body stands for widespread conditions, or wearable patches for continuous low-dose therapy.
🔹 Adapt to User Profiles: Skin-type stratification (Fitzpatrick I–VI), pediatric vs. adult dosing limits, clinical prescription modes vs. consumer wellness settings, and multilingual interfaces for global markets.
🔹 Differentiate Through Experience: Smart dose tracking, dermatologist connectivity, ergonomic form factors, quiet operation, and premium materials that elevate perceived value and justify premium pricing.
Off-the-shelf devices rarely align with these nuanced requirements. A specialized OEM private label manufacturer bridges the gap by engineering devices from the photon level up, tailored to your brand’s clinical claims, aesthetic vision, and commercial strategy.

Core Capabilities of a High-Performance OEM Private Label Partner
Not all manufacturers offer true customization. Look for partners who demonstrate these foundational competencies:
✅ Optical & Photobiological Engineering Expertise
Precision starts at the light source. Leading OEMs utilize spectrally tuned LEDs or filtered lamp arrays, paired with advanced diffusers, reflectors, and collimating optics to ensure uniform irradiance distribution. Real-time radiometric feedback and closed-loop control maintain dose accuracy across temperature fluctuations and device lifespan.
✅ Intelligent Firmware & Connectiviteit
Modern UV therapy thrives on data. Customizable firmware enables dose calculators, progressive escalation protocols, session logging, and Bluetooth/Wi-Fi connectivity. Private label app development allows brands to embed their UI, educational content, telehealth integration, and subscription services directly into the user journey.
✅ Medical-Grade Industrial Design & Materialen
Consumer acceptance hinges on aesthetics and usability. OEMs with in-house ID/MD teams select UV-stable polymers, anodized aluminum heat sinks, ergonomic grips, and intuitive touch controls. Devices are engineered for residential environments without sacrificing clinical durability.
✅ Scalable, Certified Manufacturing
From prototype to mass production, quality must remain consistent. Top-tier partners operate ISO 13485-certified facilities, implement statistical process control (SPC), conduct 100% irradiance calibration, and provide individual device test certificates. Production lines are designed for flexible batch sizes, enabling brands to validate markets before scaling.
✅ End-to-End Private Label Integration
True private label goes beyond slapping a logo on a generic box. It encompasses custom packaging design, multilingual documentation, branded accessory kits (goggles, timers, skin measurement tools), retailer-ready UPC/EAN labeling, and co-branded marketing collateral that reinforces clinical credibility.
The Private Label Advantage: Speed, Control, and Commercial Agility
Partnering with a custom OEM private label manufacturer delivers tangible business benefits:
⚡ Accelerated Time-to-Market: Leverage pre-validated optical platforms, regulatory-ready designs, and established supply chains to launch in 6–9 months instead of 18–24.
💰 Capital Efficiency: Convert fixed R&D and tooling costs into variable production expenses. Pay only for what you sell, with flexible MOQs and phased scaling options.
🛡️ Brand Ownership & IP Protection: Custom firmware, unique form factors, and proprietary dosing algorithms become your brand assets. Formal NDAs, clear IP assignment clauses, and secure manufacturing protocols safeguard your innovations.
🌍 Global Market Readiness: Multi-region certification planning (FDA, CE LOL, TGA, PMDA), voltage/plug localization, and language-specific UIs enable simultaneous launches across key territories.
📊 Data-Driven Iteration: Post-launch telemetry, user feedback loops, and field performance analytics inform firmware updates and next-generation hardware revisions without disrupting current inventory.
Navigating the Custom Development Workflow
- Ontdekking & Specificatie: Joint workshops define clinical targets, regulatory pathway, target user, irradiance requirements, and budget parameters.
- Optical & Mechanical Design: Ray-tracing simulations, thermal modeling, and ergonomic prototyping yield functional alpha units.
- Firmware & App Development: Custom UI, dose algorithms, connectivity protocols, and cloud architecture are built and tested.
- Pre-Compliance Validation: In-house photobiological, electrical, EMC, and safety interlock testing de-risk formal certification submissions.
- Ondersteuning bij indiening van regelgeving: Technical file preparation, notified body coordination, and audit readiness documentation.
- Pilot Production & Field Testing: Small-batch manufacturing for clinician trials, user feedback, and final design refinement.
- Massaproductie & Launch: Scalable output, serialization, quality audits, and logistics coordination for market deployment.
- Post-Market Support: Firmware-updates, spare parts management, warranty fulfillment, and continuous improvement cycles.
Naleving & Veiligheid: Non-Negotiable Foundations for UV Devices
UV-emitting devices carry inherent photobiological risks if improperly engineered. A responsible private label manufacturer embeds safety into every design layer:
🔍 Photobiological Risk Assessment: IEC 62471 evaluation classifies devices into risk groups (typically Group 1 of 2 for home use), with mandatory labeling, protective eyewear, and exposure limits.
⚡ Electrical & Thermal Safety: Compliance with IEC 60601-1 (medisch) or IEC 60335-1 (household), redundant thermal cutoffs, fault-tolerant power supplies, and EMC/EMI shielding.
📋 Risk Management Framework: ISO 14971-aligned hazard analysis, failure mode evaluation (FMEA), and mitigation strategies documented throughout the design history file.
🌐 Data Privacy & Cybersecurity: GDPR/HIPAA-compliant data handling, encrypted cloud storage, secure OTA updates, and user consent workflows for connected devices.
📜 Regulatory Documentation: Complete technical files, klinische evaluatierapporten, test certificates, and post-market surveillance plans ready for audit submission.
Brands that prioritize compliance from day one avoid costly redesigns, market delays, and liability exposure.
Strategic Partnership vs. Transactional Sourcing: What Sets Leaders Apart
The difference between a vendor and a strategic OEM partner lies in long-term alignment:
🤝 Co-Innovation Roadmaps: Joint planning for next-generation features, new wavelength integrations, or modular accessory ecosystems that keep your brand ahead of competitors.
🤝 Supply Chain Transparency: Direct relationships with tier-1 LED suppliers, driver manufacturers, and optical component producers ensure wavelength consistency, pricing stability, and shortage resilience.
🤝 Quality Accountability: AQL-based inspection protocols, pre-shipment third-party audits, traceable batch records, and rapid root-cause analysis for field issues.
🤝 Commercial Enablement: Training programs, demo unit allocation, clinical summary sheets, retailer onboarding kits, and co-marketing support that accelerate channel adoption.
🤝 IP & Territory Protection: Clear contractual boundaries preventing manufacturers from selling identical designs to direct competitors in your designated markets.
Transaction-focused suppliers optimize for lowest unit cost; strategic partners optimize for brand longevity, klinische geloofwaardigheid, and market share growth.
Positioning Your Private Label UV Therapy Brand for Market Leadership
Product development is only half the equation. Successful brands leverage their OEM partnership to build defensible market positioning:
🎯 Clinical Credibility: Publish pilot data, secure dermatologist endorsements, and emphasize dosimetry precision and safety certifications in marketing materials.
🎯 Hybrid Care Models: Integrate with telehealth platforms, offer prescription-to-home device programs, and enable remote dermatologist oversight for continuous care.
🎯 Tiered Product Architecture: Launch entry-level consumer panels alongside professional-grade clinic devices, using shared core technology to maximize R ROI.
🎯 Subscription & Service Ecosystems: Bundle hardware with replacement components, skincare protocols, AI dose optimization, or premium coaching to drive recurring revenue.
🎯 Regulatory-First Messaging: Lead with compliance badges, test report transparency, and safety interlock explanations to build trust in an increasingly scrutinized market.
Conclusie: Jouw merk, Engineered for Phototherapy Leadership
The UV skin therapy market rewards brands that combine clinical precision with consumer-centric design, regulatory rigor with commercial agility, and technological innovation with brand authenticity. A custom UV skin therapy devices OEM private label manufacturer provides the technical depth, manufacturing excellence, and strategic partnership required to turn your vision into a trusted, life-enhancing product line.