The global market for at-home aesthetic devices has experienced explosive growth, with red light therapy (РЛТ) face masks leading the charge. Consumers increasingly seek clinical-grade skincare solutions that deliver visible results without frequent spa visits. Однако, not all LED masks are created equal. For brands, distributors, and clinics sourcing these devices, partnering with an FDA CE certified red light therapy face mask manufacturer specializing in high irradiance output is no longer optional—it’s essential for safety, эффективность, and market credibility.
Why Certification Is Non-Negotiable in Medical-Grade Skincare
Regulatory certifications like FDA 510(к) clearance and CE marking under MDR (Medical Device Regulation) serve as critical validation that a device meets rigorous standards for safety, performance, and quality management. An FDA-cleared red light therapy mask has undergone review demonstrating substantial equivalence to legally marketed predicate devices, with documented evidence of:
- Photobiological safety (МЭК 62471): Ensuring emitted light poses no risk to eyes or skin
- Electrical safety (МЭК 60601-1): Critical for devices used near the face with lithium batteries
- Biocompatibility (ИСО 10993): Confirming skin-contact materials are non-irritating and hypoallergenic
- Clinical performance data: Supporting claimed benefits like wrinkle reduction or acne improvement
Similarly, CE certification under the EU’s stricter MDR framework requires comprehensive technical documentation, post-market surveillance plans, and adherence to ISO 13485 quality management systems. Для B2B-покупателей, verifying a manufacturer’s certifications isn’t just due diligence—it’s risk mitigation. Uncertified devices may face customs seizures, liability exposure, or reputational damage if adverse events occur.
What Does “High Irradiance” Really Mean?
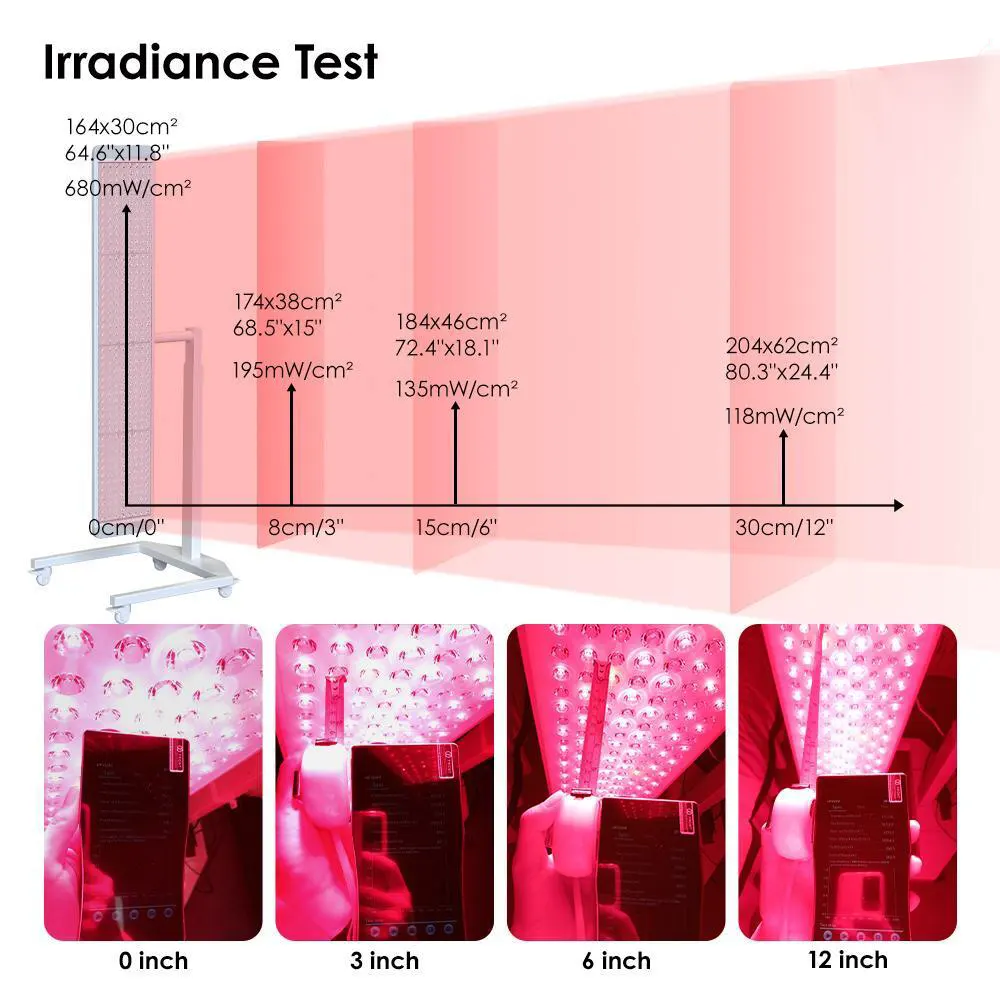
“Irradiance” refers to the power of light energy delivered to the skin, typically measured in мВт/см² (milliwatts per square centimeter).
Why Irradiance Matters
Higher irradiance means:
- deeper skin penetration
- faster biological response
- shorter treatment time
- more visible results
Professional vs Consumer-Level Output
| Device Type | Irradiance Level |
|---|---|
| Low-end masks | 10–30 mW/cm² |
| Mid-range devices | 30–80 mW/cm² |
| Professional-grade masks | ≥80–120 mW/cm² |
👉 For B2B buyers, ≥80 mW/cm² is the minimum benchmark for serious skincare performance.
The Science Behind High Irradiance: Why mW/cm² Determines Real Results
- Minimum threshold: ≥20 mW/cm² at the skin surface for meaningful mitochondrial activation
- Optimal range: 30–50 mW/cm² for balanced penetration and safety in facial applications
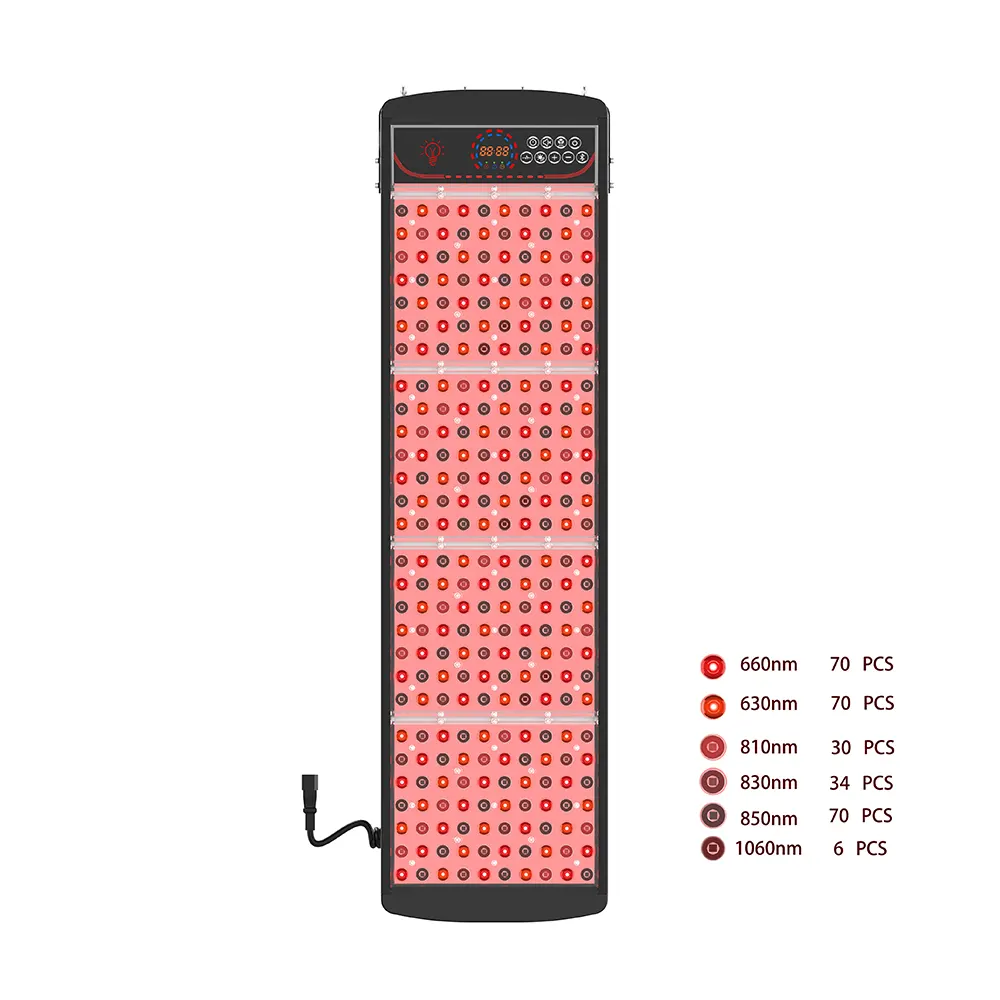
- Wavelength precision: 630–660 нм (красный) for superficial collagen synthesis + 810–880nm (ближний инфракрасный) for deep tissue repair
- Strategic LED placement: Ensuring uniform coverage without shadow zones
- Medical-grade lenses: Focusing light output rather than scattering it
- Thermal management: Preventing LED efficiency drop-off during use
- Calibrated drivers: Maintaining stable output throughout the treatment cycle
What to Evaluate in a High-Performance Manufacturer
When sourcing FDA CE certified high-irradiance masks, assess potential partners across four critical dimensions:
1. Technical R&D Capability
- In-house optical engineering team for wavelength calibration and irradiance mapping
- Proprietary silicone molding processes ensuring 98% facial coverage and light seal
- Firmware development for smart features: pulse modulation (например, 10Hz for enhanced ATP production), app connectivity, usage tracking
2. Regulatory Expertise
- Proven track record securing FDA 510(к), CE MDR, ТГА (Австралия), and other key market approvals
- Dedicated regulatory affairs staff to manage documentation, testing, and submissions
- Experience with combination products (например, masks with integrated sensors or AI skin analysis)
3. Quality & Масштабируемость
- ИСО 13485 certified production facility with cleanroom assembly for optical components
- Rigorous incoming QC: LED binning for wavelength consistency (±5nm tolerance), battery safety testing
- Flexible MOQs and OEM/ODM support for custom wavelengths, branding, or packaging
4. Post-Market Support
- Comprehensive technical documentation: user manuals, service guides, risk analysis reports
- Warranty terms aligned with medical device expectations (typically 12–24 months)
- Responsive engineering support for troubleshooting or design iterations
Чистыйлюкстек: A Case Study in Certified High-Irradiance Innovation
- 66 medical-grade LEDs delivering 20±5 mW/cm² irradiance at 0cm distance
- Triple-wavelength configuration: 633нм (collagen boost) + 850нм (deep repair) + 1072нм (targeted anti-aging)
- FDA 510(к), CE, ФКС, and GB4706 certified with 2000mAh battery providing 7×10-minute sessions per charge
- Food-grade silicone construction with IP20 rating for easy sanitization
- 93 мощные светодиоды (279 chips) achieving 35–40 mW/cm² irradiance
- Five-wavelength spectrum (415/590/633/850/1072нм) supporting acne, против старения, and sensitivity protocols
- МЭК 60601-1 medical electrical safety certification alongside ISO 13485 quality management
- Foldable design with 4000mAh battery enabling 10–12 treatments per charge
B2B Buyer’s Checklist: Key Parameters for Procurement
✅ Wavelength accuracy certificate: Spectroradiometer confirmation of peak wavelengths within ±5nm tolerance
✅ Certification copies: Current FDA 510(к) letter, CE Declaration of Conformity, ИСО 13485 certificate
✅ Material safety documentation: ИСО 10993 biocompatibility reports for all skin-contact components
✅ Battery safety compliance: UN38.3, МЭК 62133, or equivalent certification for lithium cells
✅ Thermal performance data: Surface temperature measurements during continuous operation
✅ Usage protocol guidance: Evidence-based recommendations for treatment duration and frequency
Заключение: Investing in Credibility and Efficacy
- Regulatory confidence: Streamlined market entry across North America, Europe, and other regulated regions
- Clinical credibility: Devices capable of delivering the irradiance levels validated in peer-reviewed PBM research
- Brand differentiation: Premium positioning through verifiable performance metrics and safety standards
- Long-term partnership: Engineering support for iterative innovation as technology and regulations evolve