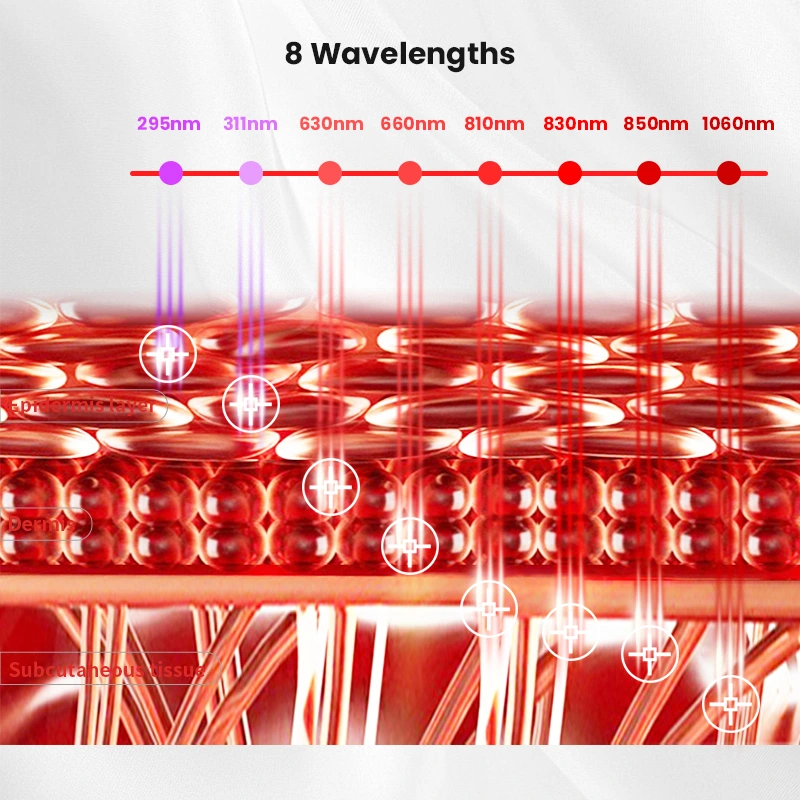
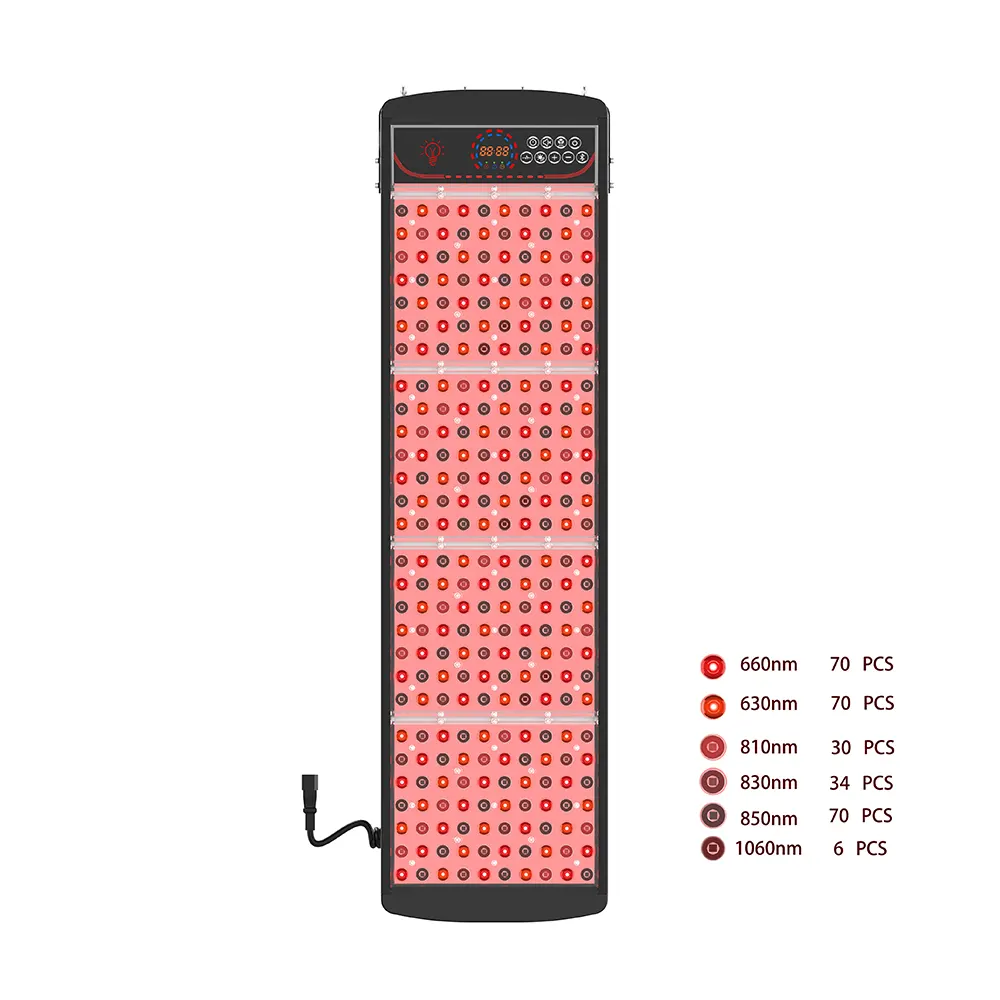
The rapid expansion of the at-home medical device market has created a significant opportunity for distributors specializing in dermatological wellness solutions. Among the fastest-growing categories is UVB light therapy equipment for home treatment—a clinically validated modality for managing psoriasis, vitiligo, eczema, and other chronic skin conditions.
Per i distributori, securing reliable, high-quality inventory through bulk purchasing is essential to meeting rising consumer demand, achieving competitive pricing, and building long-term brand partnerships. This article provides a strategic framework for distributors evaluating bulk procurement of home-use UVB therapy devices, covering supplier selection, quality assurance, logistics optimization, and go-to-market execution.
The Distributor Advantage: Why Bulk UVB Procurement Makes Strategic Sense
Distributors occupy a critical position in the UVB therapy value chain—connecting manufacturers with clinics, retailers, telehealth platforms, and direct-to-consumer channels. Bulk purchasing offers distinct advantages:
📈 Margin Optimization: Volume-based pricing from OEM suppliers typically reduces unit costs by 25–45%, enabling healthier margins or more aggressive retail positioning.
📦 Inventory Stability: Securing committed production slots ensures product availability during peak demand seasons (per esempio., winter flare-ups for psoriasis patients) and mitigates supply chain disruptions.
🤝 Brand Partnership Leverage: Large-volume commitments often unlock preferential terms: extended payment windows, co-marketing support, exclusive regional rights, or early access to new product launches.
🚀 Ingresso sul mercato più rapido: With pre-vetted, certified inventory ready to ship, distributors can accelerate onboarding of new retail partners or launch DTC campaigns without lengthy lead times.
Tuttavia, these benefits depend entirely on selecting the right manufacturing partner and structuring procurement agreements that protect quality, conformità, and brand reputation.
Key Criteria for Evaluating UVB Equipment Suppliers for Bulk Orders
Not all manufacturers are equipped to support distributor-scale procurement. Prioritize suppliers who demonstrate:
✅ Medical-Grade Certification Portfolio
Home-use UVB devices straddle the line between wellness gadget and medical device. Ensure your supplier holds relevant certifications for your target markets:
- Registrazione FDA & Listing (U.S.A.)
- CE Marking under MDR or as a Class I/IIa medical device (Unione Europea)
- CEI 62471 Photobiological Safety Report
- CEI 60601-1 O CEI 60335-1 Electrical Safety Compliance
- ISO 13485 Quality Management System Certification
Request copies of test reports and certificates during supplier qualification—not after order placement.
✅ Scalable, Transparent Production Capacity
Verify the supplier’s ability to fulfill your volume requirements without compromising quality:
- Monthly production capacity and current utilization rates
- Lead times for standard vs. customized configurations
- Component sourcing strategy (direct vs. brokered) for critical parts like UVB LEDs
- Contingency plans for raw material shortages or logistics delays
A reputable supplier will provide factory audit reports, production schedules, and real-time order tracking.
✅ Consistent Optical Performance & Controllo di qualità
UVB therapeutic efficacy depends on precise wavelength output (typically narrowband 311 nm) and uniform irradiance. Demand evidence of:
- Incoming LED binning protocols to ensure wavelength consistency (±3 nm tolerance)
- 100% final irradiance calibration with individual device certificates
- Aging tests to validate output stability over claimed lifespan (per esempio., 10,000+ ore)
- Statistical process control (SPC) data for key performance parameters
Suppliers unwilling to share quality metrics pose significant reputational risk.
✅ Flexible Customization & Private Label Support
Distributors often need to differentiate offerings across channels. Look for suppliers offering:
- Private-label hardware with your branding, confezione, and UI language
- Modular configurations (per esempio., panel size, irradiance level, accessory bundles)
- Firmware customization for region-specific safety limits or app integration
- MOQ flexibility for pilot orders before scaling to full bulk purchases
Structuring Your Bulk Purchase Agreement: Protecting Your Investment
A well-negotiated contract is as critical as product quality. Key clauses to include:
🔹 Quality Acceptance Criteria: Define measurable pass/fail standards for irradiance, precisione della lunghezza d'onda, cosmetic defects, and functional testing. Specify inspection protocols (per esempio., AQL 1.0 for critical defects).
🔹 Garanzia & Supporto post-vendita: Clarify warranty duration (typically 1–3 years), coverage scope (parts/labor), and return/repair logistics. Prefer suppliers offering regional service hubs or advance replacement programs.
🔹 Intellectual Property & Brand Protection: Ensure clear ownership of custom designs, firmware, and marketing assets. Include confidentiality clauses and restrictions on supplier selling identical products to competing distributors in your territory.
🔹 Pricing Escalation & Volume Tiers: Lock in pricing for initial orders while establishing transparent adjustment mechanisms for future batches (per esempio., tied to LED index prices). Negotiate volume discounts that reward growth.
🔹 Regulatory Responsibility Allocation: Define which party manages certification updates, post-market surveillance, and adverse event reporting—critical for medical-adjacent products.
Engage legal counsel experienced in medical device distribution to review agreements before signing.
Logistica, Warehousing, and Inventory Management for UVB Devices
Bulk purchasing introduces operational complexities that require proactive planning:
📦 Temperature-Controlled Shipping: UVB LEDs and electronic components can degrade under extreme temperatures. Specify climate-controlled logistics for ocean/air freight and last-mile delivery.
🏭 Strategic Warehousing: Position inventory in regional hubs to reduce shipping times and costs. Consider third-party logistics (3PL) partners with experience handling medical-adjacent electronics.
🔄 Inventory Turnover Planning: UVB devices have shelf-life considerations (per esempio., LED lumen depreciation). Implement FIFO (first-in, first-out) systems and align purchase volumes with realistic sales forecasts to avoid obsolescence.
📋 Serial Number Traceability: Maintain batch-level tracking for warranty claims, recalls, or regulatory inquiries. Require suppliers to provide unique serial numbers and manufacturing date codes on each unit.
Go-to-Market Support: Maximizing ROI on Bulk Inventory
Procurement is only the first step. Partner with suppliers who actively support your commercial success:
🎯 Marketing Collateral: Request high-resolution product imagery, clinical summary sheets, comparison guides, and demo units for retailer training or influencer campaigns.
🎯 Technical Training: Ensure your sales and support teams receive comprehensive product education—wavelength science, dosing protocols, safety guidelines, and troubleshooting.
🎯 Channel Strategy Alignment: Collaborate on positioning for different outlets: clinical distributors may emphasize FDA clearance and dosimetry precision, while retail partners prioritize ease of use and aesthetic design.
🎯 Data-Driven Reordering: Share sell-through data with your supplier to optimize production planning and secure priority allocation for high-demand SKUs.
Risk Mitigation: Safeguarding Your Distribution Business
Bulk purchasing concentrates risk. Implement these safeguards:
⚠️ Diversify Supplier Base: Avoid over-reliance on a single manufacturer. Qualify 2–3 backup suppliers to ensure continuity during disruptions.
⚠️ Pre-Shipment Inspections: Engage third-party quality inspectors (per esempio., SGS, Bureau Veritas) to verify product conformity before final payment and shipment.
⚠️ Regulatory Monitoring: Track evolving regulations in your target markets (per esempio., EU MDR transitions, FDA guidance updates) and ensure your supplier adapts proactively.
⚠️ Insurance Coverage: Secure product liability insurance that covers UV-emitting devices, with clear definitions of coverage scope and claim procedures.
Building Long-Term Partnerships: Beyond Transactional Procurement
The most successful distributors view suppliers as strategic allies, not just vendors. Cultivate partnerships through:
🤝 Joint Business Planning: Share market insights, sales forecasts, and customer feedback to co-develop product roadmaps and promotional calendars.
🤝 Co-Innovation Opportunities: Collaborate on next-generation features—smart connectivity, AI dose optimization, or modular accessories—that differentiate your offerings.
🤝 Performance-Based Incentives: Structure agreements that reward mutual growth, such as rebates for exceeding volume targets or shared investment in market development.
Conclusione: Strategic Bulk Sourcing as Your Competitive Edge
Per i distributori, bulk purchasing of home-use UVB light therapy equipment is more than a procurement decision—it’s a strategic lever for market leadership. By partnering with a certified, scalable, and collaborative manufacturer, you secure the quality, conformità, and commercial support needed to capitalize on one of dermatology’s fastest-growing segments.
Success hinges on rigorous supplier qualification, thoughtful contract structuring, operational excellence in logistics, and proactive go-to-market execution. When executed strategically, bulk procurement transforms inventory investment into sustainable competitive advantage—enabling you to deliver clinically meaningful solutions to consumers while building a resilient, high-growth distribution business.