ウェルネステクノロジーの状況は急速に進化しています, と 赤色光療法 (光生体調節) ニッチなクリニックから主流の家庭用への移行. 消費者の意識が高まるにつれて, 安全に対する要求も同様です, 効果的, および法的に準拠したデバイス. この有利な市場への参入を検討しているブランド向け, と提携している FDA が赤色光療法パネルの製造業者を認可 それは単なる好みではなく、戦略的な必要性です.
北米の規制環境を乗り切るのは複雑な場合があります. 米国. 食品医薬品局 (FDA) ユーザーの安全を確保するために、医療機器およびウェルネス機器に対して厳格な基準を設定しています。. 適切な通関手続きなしにメーカーを選択すると、税関で差し押さえられる可能性があります, 法的責任, そして取り返しのつかないブランドのダメージ.
この包括的なガイドでは、FDA 認可の重要性について説明します。, メーカーの認証情報を確認する方法, そしてなぜ ピュアラックステック コンプライアンスを求めるブランドにとって信頼できるパートナーとして傑出しています, 高性能光療法ソリューション.

赤色光治療装置に関して FDA 認可が重要な理由
用語 “FDAの承認済み” 世界市場で大きな比重を占めている, 特に米国では. これは、デバイスが審査を受けており、合法的に販売されている述語デバイスと実質的に同等であるとみなされることを示します。.
1. 市場アクセス & 信頼
アマゾンのような小売業者, ウォルマート, 専門的な流通チャネルでは、健康関連機器を出品する前に FDA 認可の証明が必要になることがよくあります。. さらに, 消費者の教育はますます進んでいます; 彼らは安全性と有効性の証として認証マークを求めます.
2. 法的保護
準拠していない医療機器や健康機器を販売すると、警告書が発行される可能性があります, 罰金, または製品リコール. アン FDA が赤色光療法パネルの製造業者を認可 あなたのビジネスを規制執行から守るために必要な文書を提供します.
3. 保険 & 責任
製造物賠償責任保険会社の多くは、保険契約を発行する前に規制遵守の証拠を要求しています。. FDA の認可は、設計段階と製造段階でデューデリジェンスが実施されたことを証明します.
4. 臨床的信頼性
カイロプラクターをターゲットとするブランド向け, 理学療法士, またはメディスパ, 多くの場合、FDA の認可が調達の前提条件となります. デバイスが安全性と出力に関する特定の性能基準を満たしていることを検証します。.
FDAを理解する 510(k) 対. FDA登録済み
FDA の相互作用のさまざまなレベルを区別することが重要です, 多くのメーカーがこれらの用語を誤用しているため、.
- FDA登録済み: これは単に、その施設が FDA に登録されていることを意味します。. それはあります ない 製品自体が安全性について審査または認可されていることを意味します.
- FDA 510(k) クリア済み: これは、特定のデバイス モデルが市販前通知を受けており、述語デバイスと比較した安全性と有効性に基づいてマーケティングが許可されていることを示します。.
を検索するときは、 FDA が赤色光療法パネルの製造業者を認可, 確実に提供できるようにする 510(k) 許可番号 (例えば, Kxxxxxx) 販売しようとしている特定の製品について. Pureluxtech は次のポートフォリオを維持しています。 510(k)-クリアされたデバイス, パートナーが自信を持ってマーケティングできるようにする.
真のFDA認可を受けた製造業者の定義とは?
コンプライアンスを主張するすべての工場が合法であるわけではない. 本当の FDA が赤色光療法パネルの製造業者を認可 生産プロセス全体を通じて厳格な品質管理システムを遵守します.
1. ISO 13485 認証
FDA は ISO との整合性を模索することがよくあります 13485, 医療機器の品質管理システムの国際規格. これにより設計の一貫性が確保されます, 発達, 生産, そしてインストール.
2. 設計履歴ファイル (DHF)
準拠したメーカーは、デバイスの設計プロセスの詳細な記録を保持しています。, リスク分析も含めて, 検証, そして検証. この文書は FDA 監査の際に重要です.
3. コンポーネントのトレーサビリティ
あらゆるLEDチップ, ドライバ, ハウジングコンポーネントは追跡可能でなければなりません. リコールや品質問題が発生した場合, メーカーは影響を受けるバッチを迅速に特定できなければなりません.
4. 市販後の監視
コンプライアンスは出荷時に終わりではありません. 責任あるメーカーが現場のパフォーマンスを監視し、必要に応じて有害事象を報告します。, 継続的な安全の確保.
ピュアラックステック: 信頼できる FDA の認可を受けた赤色光療法パネルのメーカー
に設立 2015 そして本社は深センにあります, ピュアラックステック 準拠した光療法製造のリーダーとしての地位を確立しました. 10年近くの経験を活かして, 私たちは世界的な規制の微妙な違いを理解しています, 特に米国の厳しい要件. 市場.
当社のコンプライアンス認証情報
- FDA 510(k) クリアされたデバイス: 当社のパネルとマスクのポートフォリオの特定のモデルは、 510(k) プロセス.
- ISO 13485 認定済み: 当社の品質管理システムは国際医療機器基準を満たしています.
- 追加の認定: CE, UKCA, FCC, RoHS, 到着, とTGA (オーストラリア), 世界市場への準備を確実にする.
- SGS工場監査: 当社の施設は、生産基準を検証するために定期的に第三者監査を受けています。.
卓越した製造
コンプライアンスは生産ラインに組み込まれています. ピュアラックステックが運営 10 自動SMT (表面実装技術) 行, LEDの正確な配置と一貫したはんだ付け品質を確保. すべてのユニットは、 6-ステップ品質管理プロセス, 含む:
- IQ: すべての原材料の受入品質管理.
- SMT検査: 自動光学検査 (あおい) PCB組み立て中.
- 組立チェック: 構造的完全性の検証.
- 老化試験: 48-1時間の連続運転で初期故障を特定.
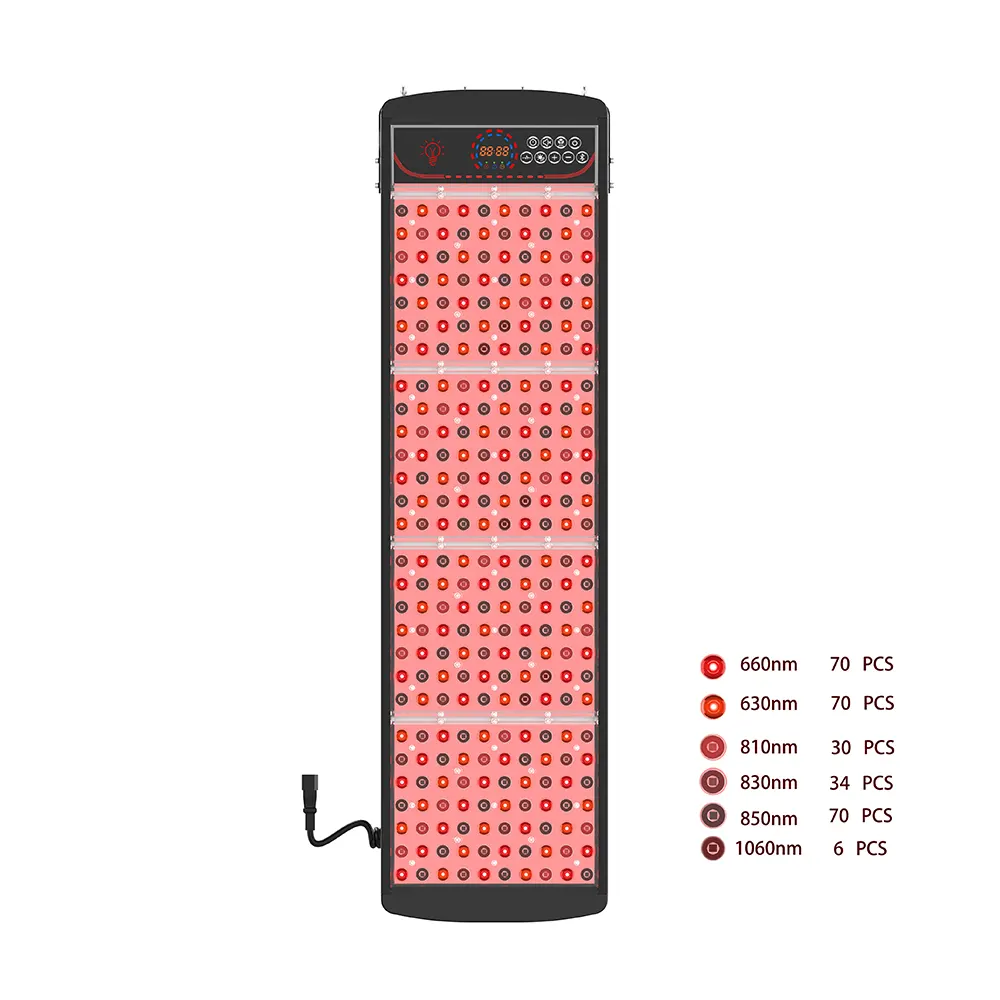
- 機能テスト: 波長出力の検証 (660nm/850nm) と放射照度.
- 最終監査: 包装およびラベルの適合性チェック.
この厳格なプロセスにより、すべての FDA が赤色光療法パネルを認可 私たちの施設を離れることは最高の安全性とパフォーマンス基準を満たしています.
🔹 OEM/ODM サービス: ブランドにとってコンプライアンスが簡単に
多くのブランドにとって, FDAの手続きを一人で進めるのは気が遠くなる. Pureluxtech は、包括的なサービスを通じてこれを簡素化します。 OEM/ODMサービス. 私たちはただ製造するだけではありません; 私たちはお客様と提携して、お客様のブランドが確実にコンプライアンスを遵守できるようにします.
1. 事前にクリアされたプラットフォーム
すでに FDA の認可を受けた既存のデバイス プラットフォームを提供します. これらのデザインを活用することで、, 市場投入までの時間と規制コストを大幅に削減できます.
2. ドキュメントのサポート
必要な技術ファイルを提供します, テストレポート, ブランドの登録と通関に必要な認証書類.
3. ラベリング & パッケージングのレビュー
FDA の規制により、特定のラベル表示要件が定められています (例えば, 使用目的, 警告, メーカー情報). 私たちのチームは、製造前にパッケージアートワークをレビューして、規制ガイドラインに準拠していることを確認します.
4. 妥協のないカスタマイズ
ハウジングをカスタマイズできる, ブランディング, ソフトウェア, コンプライアンスを犠牲にすることなく均一な波長比を実現. 私たちのR&Dチームの 200+ エンジニア あらゆる変更がクリアランスに必要な安全プロファイルを維持することを保証します.
製品ポートフォリオ: あらゆるニーズに対応する認定ソリューション
Pureluxtech はさまざまな製品を提供しています。 FDA が赤色光療法パネルを認可 さまざまな市場セグメントに適した関連デバイス.
|
製品シリーズ
|
説明
|
理想的な市場
|
|---|---|---|
|
MAXPRO パネル
|
ハイパワー, 均一な放射照度を備えたプロ仕様のパネル.
|
クリニック, メドスパス, ジム
|
|
NOVAパネル
|
中型, ホームウェルネス愛好家向けの多用途パネル.
|
DTC ブランド, 小売り
|
|
コンパクトパネル
|
ポータブル, 目的を絞った治療のための旅行に適したデザイン.
|
トラベルウェルネス, 法人ギフト
|
|
LED 治療用マスク
|
顔のスキンケアとアンチエイジングのための FDA 認可済みのオプション.
|
ビューティーブランド, スキンケアライン
|
|
育毛ヘルメット
|
臨床的に研究された育毛用の波長.
|
毛髪学, 男性/女性の健康
|
|
ペット治療装置
|
動物の健康のために設計された安全な波長.
|
獣医, ペットケア ブランド
|
各製品は過熱保護などの安全機能を備えて設計されています。, 目の安全に関するコンプライアンス, 耐久性のある住宅材料.
FDA 認可に関するよくある誤解
ブランドが情報に基づいた意思決定を行えるよう支援する, いくつかの一般的な通説を明確にしましょう:
- 神話: “FDAの承認” と同じです “FDAの認可を受けています。”
- 事実: ほとんどの光線療法装置は、 510(k) クリア済み, ない “承認された。” 承認は高リスクのクラス III デバイスに留保されています. クリアは安全で既存のデバイスと同等であることを意味します.
- 神話: どの LED ライトも安全に販売できます.
- 事実: 不適切な波長や過剰な照射は目の損傷や皮膚の火傷を引き起こす可能性があります. コンプライアンスにより、出力が安全な制限内に収まることが保証されます.
- 神話: クリアしたら, 常にクリアされています.
- 事実: 大幅な設計変更には、新しいものが必要になる場合があります。 510(k) 提出. Pureluxtech は、変更が規制当局の審査を引き起こすタイミングについてパートナーにアドバイスします.
Pureluxtech と提携してコンプライアンスに準拠した成長を実現
赤色光療法市場への参入は計り知れない可能性をもたらします, ただし、コンプライアンスと品質の基盤に基づいて構築された場合に限ります. 主役として FDA が赤色光療法パネルの製造業者を認可, Pureluxtech は専門知識を提供します, インフラストラクチャー, あなたのブランドが世界的に成功するためのサポートを提供します.
Pureluxtechの違いをぜひ体験してください。. 初期コンセプトから最終納品まで, 私たちは安全な製品であなたのブランドを強化することに全力で取り組んでいます, 効果的, および認定された光療法ソリューション.
✅ 準拠製品への取り組みを開始しましょう
- 資格情報の検証: FDA および ISO 認証文書をリクエストしてください.
- サンプルのリクエスト: パネルの品質と放射照度出力を直接テストしてください.
- 専門家に相談する: ターゲット市場とコンプライアンスのニーズについて当社のチームと話し合ってください。.
よくある質問 (よくある質問)
Q1: 自分自身の FDA 登録なしで Pureluxtech パネルを米国で販売できますか? あ: 私たちのデバイスがクリアされている間、, 米国で医療機器を販売するブランドは通常、その施設を登録し、FDAに機器をリストする必要があります. 登録をサポートするために必要な書類を提供します.
第2四半期: 通関のためのFDA文書を提供していますか?? あ: はい, FDAを含む包括的なコンプライアンスパッケージを提供します 510(k) 参考文献, CE証明書, スムーズな通関を促進するためのテストレポート.
Q3: カスタム OEM パネルの承認を得るまでにどのくらい時間がかかりますか? あ: 当社の既存のクリア済みプラットフォームを使用している場合, はるかに速く起動できます. 新しいカスタム デザインには追加のテストと提出時間が必要になる場合があります, 私たちのチームが管理を支援できるもの.
Q4: パネルは目にさらされても安全ですか? あ: 当社のパネルはレーザーの安全基準を満たすように設計されています (IEC 62471). しかし, 治療中は付属の保護メガネを使用することをお勧めします, 特に高出力近赤外線モデルの場合.
Q5: FDA の認可を受けたデバイスの保証ポリシーは何ですか? あ: 当社では以下の標準保証を提供しています。 1 に 3 年. 当社の品質管理プロセスは低い故障率を保証します, ただし、保証請求を効率的にサポートするためにスペアパーツの在庫を維持しています.