The at-home hair restoration sector has transitioned from a niche wellness trend into a multi-billion-dollar mainstream category. Consumers across the United States and Europe are actively seeking non-invasive, clinically backed alternatives to pharmaceutical treatments and surgical procedures. 저수준 레이저 치료 (LLLT) 장치, particularly red light hair growth caps, sit at the center of this demand surge. 아직, despite rising consumer interest, many regional distributors, retail chains, clinic networks, and e-commerce aggregators struggle to source reliable, compliant, and brand-ready inventory that meets Western regulatory standards and margin expectations.
Pureluxtech has engineered a dedicated distribution framework to close this gap. Our red light hair growth cap distributor opportunities USA Europe program is designed for established and emerging partners who want to capitalize on the hair wellness boom with certified hardware, competitive wholesale structures, territorial support, and end-to-end commercial enablement. This guide outlines why the US and EU markets are primed for growth, what Pureluxtech delivers to authorized distributors, and how you can secure a strategic partnership that scales with demand.

Why the USA & Europe Represent Prime Distribution Markets
🇺🇸 United States: High Demand, Clear Regulatory Pathways
The US hair care and wellness technology market continues to expand at a double-digit CAGR, driven by aging demographics, rising disposable income, and strong DTC e-commerce adoption. American consumers increasingly prefer FDA-cleared or FDA-aligned devices that offer transparent safety data and measurable outcomes. Retail channels ranging from specialty beauty stores and pharmacy chains to Amazon Professional and telehealth platforms are actively onboarding LLLT products. Distributors who can supply compliant, consistently performing inventory with reliable fulfillment hold a distinct competitive advantage.
🇪🇺 Europe: Premium Positioning & Cross-Border Scalability
European buyers prioritize clinical credibility, sustainability, and regulatory transparency. The CE marking framework, combined with growing consumer awareness of photobiomodulation, has created a structured, high-trust marketplace across the UK, 독일, France, Scandinavia, and Southern Europe. Professional trichology clinics, dermatology networks, and premium wellness retailers are expanding their device offerings, while cross-border e-commerce enables rapid regional scaling. Distributors with CE-compliant, multilingual-ready products can efficiently penetrate multiple countries without fragmented sourcing.
Both markets reward partners who combine product reliability with commercial agility. Pureluxtech’s distribution model is built specifically for this environment.
What Pureluxtech Delivers to Authorized Distributors
We don’t just supply hardware—we equip distribution partners with a complete commercial ecosystem. Our program is structured to protect margins, accelerate sell-through, and minimize operational friction:
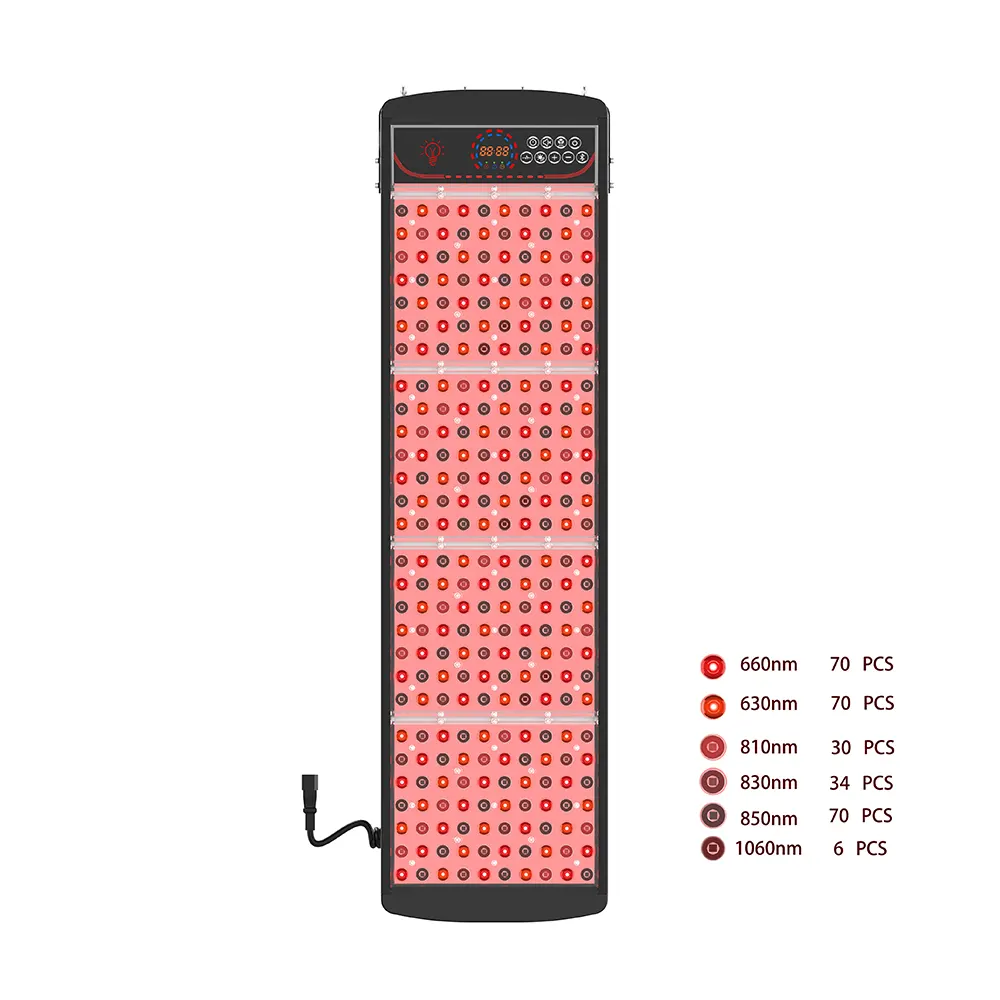
- 인증됨, Market-Ready Inventory: Every red light hair growth cap is engineered around a validated 650nm ± 5nm photobiomodulation array, featuring 120 medical-grade diodes, 5mW ± 20% Class II laser output, and integrated thermal regulation (surface ≤ 50°C). Devices ship with FDA/CE-aligned documentation, RoHS/FCC compliance, and batch-level traceability.
- Competitive Wholesale Pricing & Margin Protection: Direct factory pricing eliminates intermediary markups. Tiered volume discounts, MAP (Minimum Advertised Price) guidelines, and territorial exclusivity options protect your profitability and prevent channel conflict.
- Flexible Reorder & Inventory Planning: Start with conservative pilot allocations, then scale based on real sales velocity. Our production scheduling prioritizes authorized distributors, ensuring consistent stock availability during peak seasons or campaign launches.
- Dedicated Account & 기술지원: A single point of contact manages forecasting, compliance queries, warranty processing, 및 펌웨어 업데이트. You focus on sales; we handle supply chain complexity.
규정 준수 & Regulatory Readiness for US/EU Channels
Distributing health-adjacent technology in Western markets requires more than attractive packaging—it demands verified compliance. Pureluxtech pre-validates all distribution inventory to meet regional onboarding requirements:
- USA Market: Technical files aligned with FDA 510(케이) clearance pathways, FCC electromagnetic compatibility certification, and US-compliant labeling, 경고, and claim language. Documentation supports Amazon Professional, retail buyer reviews, and clinic procurement committees.
- European Market: CE marking under applicable directives, RoHS material compliance, UKCA readiness, 및 EU 적합성 선언 템플릿. Multilingual manuals (EN, DE, FR, ES, IT, NL, SV) and region-specific privacy/GDPR disclaimers are included standard.
- Claim & Marketing Compliance: Pre-approved product descriptions, clinical reference summaries, and disclaimer frameworks ensure your sales teams and e-commerce listings remain within regulatory boundaries while maximizing conversion potential.
This compliance-first approach drastically reduces distributor liability, accelerates retail/clinic onboarding, and builds long-term consumer trust.
Distributor Tiers & Commercial Benefits
|
Tier
|
적용 범위
|
Initial Allocation
|
Key Benefits
|
|---|---|---|---|
|
Regional Distributor
|
Specific state, province, or EU country
|
100–300 units
|
Volume pricing, co-op marketing fund, localized asset kits, priority reorder queue
|
|
National Distributor
|
Full US or single EU nation
|
300–800 units
|
Territorial protection, custom SKU options, dedicated account manager, retail onboarding support
|
|
Master/EU-Wide Partner
|
Multi-country or pan-European
|
800+ units
|
Exclusive product variants, API/app integration rights, freight consolidation, margin optimization, joint campaign planning
|
마케팅, Training & Sales Enablement
Inventory alone doesn’t drive sell-through. Pureluxtech equips distributors with comprehensive go-to-market resources:
- Retail-Ready Packaging: Double-wall cartons, magnetic gift sets, or e-commerce mailers with custom foam inserts, hygiene liners, and branded quick-start guides.
- Digital Asset Library: Studio photography, 360° 렌더링, lifestyle videos, 사양 시트, 규정 준수 인증서, and Amazon A+ content templates.
- Clinical & Sales Training: On-demand webinars covering LLLT science, 650nm wavelength benefits, objection handling, and professional channel positioning.
- E-Commerce Optimization: Listing copy, keyword strategies, review generation frameworks, and app ecosystem integration guides to boost conversion and retention.
- Co-Branded Campaign Support: Seasonal promotional templates, influencer seeding kits, and clinic demo units to accelerate market penetration.
These resources reduce your internal marketing overhead and ensure consistent, compliant messaging across all sales channels.
기호 논리학, Fulfillment & After-Sales Infrastructure
Reliable distribution requires predictable supply chains and transparent after-sales support. Pureluxtech’s operational framework is built for US/EU efficiency:
- Flexible Shipping Terms: FOB, CIF, or DDP options available. We coordinate with vetted freight forwarders for air, sea, or express delivery, with customs documentation prepared in-house.
- Regional Warehouse Coordination: For high-volume partners, we support bulk staging to US/EU third-party logistics (3PL) centers to enable 2–5 day domestic fulfillment.
- Batch Traceability & 품질관리: Every shipment includes serial logs, photometric test reports, and safety certifications. Randomized pre-shipment inspections ensure zero defect tolerance.
- 보증 & RMA Management: Streamlined replacement protocols, firmware troubleshooting guides, and dedicated technical support minimize downtime and protect end-user satisfaction.
- OTA Firmware & App Updates: Smart-enabled caps receive seamless software improvements without hardware returns, extending product lifecycle and reducing support tickets.
How to Become an Authorized Pureluxtech Distributor
Our onboarding process is designed for transparency, speed, and mutual alignment:
- 애플리케이션 & Business Verification: Submit your company profile, target channels, and regional coverage. We verify distribution capability and market fit.
- Market Plan & Tier Alignment: Review commercial terms, territorial options, and initial allocation. Select your partnership tier and compliance requirements.
- Sample Evaluation & Compliance Review: Receive production-ready units for internal testing, retail buyer presentations, or clinic validation.
- Agreement & Initial Order: Sign distribution terms, confirm pricing/MAP guidelines, and place your first allocation.
- 온보딩 & Launch Support: Access asset libraries, training modules, and account management. Coordinate freight, customs, and go-to-market timelines.
Qualified partners typically move from application to first shipment within 3–4 weeks, with expedited pathways available for time-sensitive retail or seasonal launches.